What makes up an atom quizlet
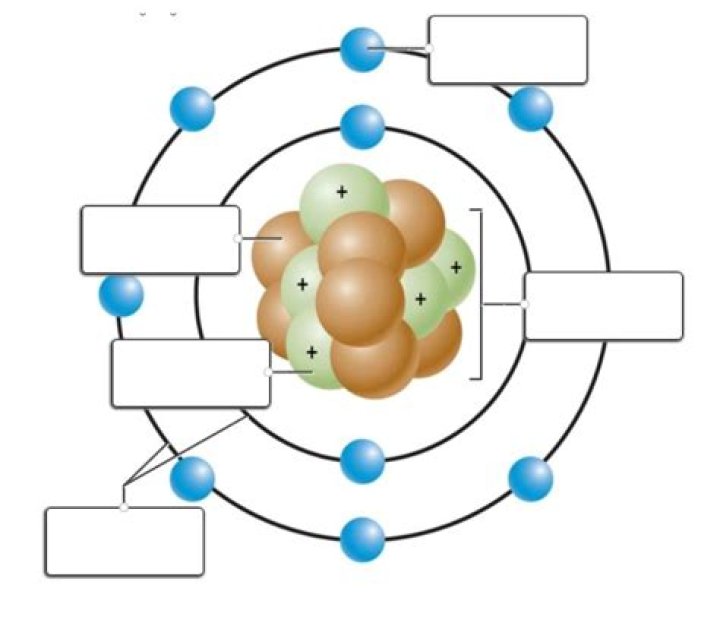
Atom. An atom is the smallest particle of an element that still has elemental properties. Atoms are made up of similar properties called protons, neutrons, and electrons. The smaller electrons orbit around a central nucleus of protons and neutrons.
What makes up a an atom?
It is composed of protons, which have a positive charge, and neutrons, which have no charge. Protons, neutrons, and the electrons surrounding them are long-lived particles present in all ordinary, naturally occurring atoms.
What are the 3 components that make up an atom?
Our current model of the atom can be broken down into three constituents parts – protons, neutron, and electrons. Each of these parts has an associated charge, with protons carrying a positive charge, electrons having a negative charge, and neutrons possessing no net charge.
What are atoms made up of quizlet?
Atoms are composed of particles called protons, electrons and neutrons. Protons carry a positive electrical charge, electrons carry a negative electrical charge and neutrons carry no electrical charge at all.What is electrons made of?
Electrons are fundamental particles so they cannot be decomposed into constituents. They are therefore not made or composed. An electron acts as a point charge and a point mass.
What is an atom in chemistry quizlet?
An atom is the smallest part of an element that can exist. Atoms are electrically neutral, with a positively charged, dense nucleus (filled with protons and neutrons) and surrounded by one or more electrons. Nucleus. the dense, positively charged mass located in the centre of an atom.
What determines the type of atom?
The number of protons in the nucleus determines which element an atom is, while the number of electrons surrounding the nucleus determines which kind of reactions the atom will undergo.
What are the 3 main parts of an atom and what do they do?
Atoms consist of three basic particles: protons, electrons, and neutrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).What determines atom interaction?
The interactions are determined by the number or electrons in the valence shell, which affects chemical behavior. Valence electrons are important, because the determine if the atom will gain electrons or give them away. … Ionic bonds consist of electrons being taken away from an atom and given to another.
What are the 4 components of an atom?Atom is made up of 4 parts, nucleus, protons, electrons and neutrons.
Article first time published onWhat are the 5 components of an atom?
- Protons.
- Neutrons.
- electrons.
Can you make a atom?
In atomic physics, scientists can create an atom, known as a Rydberg atom, in which one single electron is highly excited and orbits its nucleus at a very large distance.
What is an atom for kids?
The atom is the basic building block for all matter in the universe. Atoms are extremely small and are made up of a few even smaller particles. The basic particles that make up an atom are electrons, protons, and neutrons. Atoms fit together with other atoms to make up matter.
What is an atom easy definition?
An atom is a particle of matter that uniquely defines achemical element. An atom consists of a central nucleus that is usually surrounded by one or more electrons. Each electron is negatively charged. The nucleus is positively charged, and contains one or more relatively heavy particles known as protons and neutrons.
Are atoms made of photons?
We learn in school that matter is made of atoms and that atoms are made of smaller ingredients: protons, neutrons and electrons. Protons and neutrons are made of quarks, but electrons aren’t. … It’s not just matter: light is also made of particles called photons.
Are waves made of particles?
They’re not literally small, subatomic particles, but they act like particles when they hit other things.. Many physical interactions can be described simply as particles bouncing off of one another. On the other hand, waves are almost completely different. They’re not localized.
How are protons made?
Protons are made of fundamental particles called quarks and gluons. … Two of the quarks are called up quarks (u), and the third quark is called a down quark (d). The gluons carry the strong nuclear force between the quarks, binding them together.
Which is a characteristic of the part of the atom marked a?
An atomic nucleus contains most of the atom’s mass. Which is a characteristic of the part of the atom marked “A”? It is very dense.
How is a molecule formed quizlet?
Atoms combine to form molecules, which are substances made up of two or more atoms. What is a chemical bond? When two atoms form a molecule, the individual atoms interact in what is called a chemical bond, a form of attraction between atoms that holds them together.
What is molecule quizlet?
Molecule. A molecule is made up of two or more atoms chemically joined together. Compounds. Two or more elements are chemically joined together.
What is the main component of atoms that determines the type of bond?
The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons.
What determines how many bonds an atom will form?
The number of bonds that a particular atom will form is based on the atom’s valence electrons. Carbon for instance, which has four valence electrons, will form four bonds when it is in a molecule, as you can see from the diagram of methane below.
What is formed when an atom loses electrons?
An atom loses electrons to form a cation , that is a positively charged ion (and one that is attracted towards the negatively charged terminal, the cathode ). Both charge and mass have been conserved.
What are the 3 subatomic particles quizlet?
Protons, neutrons, and electrons are the three main subatomic particles found in an atom.
Where are the 3 parts of an atom located?
The three parts of an atom are the proton, neutron, and electron. These are the subatomic particles that interact to form the atom. Protons and neutrons are found in the nucleus at the center of the atom. While neutrons have a neutral charge, protons have a positive charge.
Where are the three main parts of an atom located?
The three main parts of a atom is protons,neutrons,and electrons. Protons – have a positive charge, located in the nucleus, Protons and neutrons have nearly the same mass while electrons are much less massive. Neutrons- Have a negative charge, located in the nucleus.
What is not made of atoms?
Well most of the things out there are not made of atoms. All fundamental particles electrons, quarks, neutrinos, gluons, bosons etc are not made of made of atoms, but it’s rather the other way around.
Is an atom an element?
AtomsElementsThe smallest part of an element.A Basic substance that can be simplified.Made of electrons, protons and neutrons.Made from one type of atom.Contain only one nucleus with numerous electrons, protons and neutrons.Atoms have the same numbers of protons.
What materials do you need to make an atom model?
You will need glue, scissors, cardstock, string, 40 large craft balls (20 of one color for the protons and the other 20 for the neutrons that’s a different color), and 20 small craft balls for the electrons.



