What products is sulfuric acid found in
The major use of sulfuric acid is in the production of fertilizers, e.g., superphosphate of lime and ammonium sulfate. It is widely used in the manufacture of chemicals, e.g., in making hydrochloric acid, nitric acid, sulfate salts, synthetic detergents, dyes and pigments, explosives, and drugs.
What products contains sulfuric acid?
Sulfuric acid can be found in many car batteries (lead-acid batteries) and in household products like drain and toilet bowl cleaners.
What household items contain Sulphuric acid?
One of the most common examples of sulfuric acid in the home is drain cleaner; liquid-form drain cleaners that unclog drains often contain sulfuric acid. In lesser concentrations, sulfuric acid occurs in glass-cleaning etching compounds, rust and corrosion dissolvers and some fabric cleaners.
What is sulfuric acid found in?
Sulfuric acid is found in battery acid and in Earth’s acid rain.Where is Sulphuric acid found at home?
Sulfuric acid is commonly found in household cleaning products, such as products to clean aluminum, though it’s not limited to that use. The reason that sulfuric acid household products are so common has to do with its corrosive properties.
What is the most common use for sulfuric acid?
Sulfuric acid is also a key substance in the chemical industry. It is most commonly used in fertilizer manufacture, but is also important in mineral processing, oil refining, wastewater processing, and chemical synthesis.
Is there Sulphuric acid in detergent?
Sulfuric acid is one of the most important industrial chemicals. … It is widely used in the manufacture of chemicals, e.g., in making hydrochloric acid, nitric acid, sulfate salts, synthetic detergents, dyes and pigments, explosives, and drugs.
Do batteries contain sulfuric acid?
Battery acid is sulfuric acid that has been diluted with water to attain a 37% concentration level. This particular type of acid is used in sealed lead acid batteries, however, concentration levels differenciate with some brands. … It is illegal to dispose of these batteries in the garbage.How can I make Sulphur at home?
Put 12.9 grams of sodium thiosulphate in a beaker and dissolve it in minimum amount of water. Pour about 15ml of nitric acid in to the beaker. Let it sit in a warm place for a couple of hours and the sulphur will settle at the bottom of the beaker. That’s it!
What drain cleaner has sulfuric acid?In the United States, you can buy sulfuric acid drain cleaner in big box stores under brand names such as Kleen-Out, Clean Shot and Liquid Lightning. These are 93 to 95 percent sulfuric acid solutions, which means they’re highly concentrated, so you must treat them with respect.
Article first time published onWhat is sulfuric acid used for in cars?
Inside your car battery is a substance called sulfuric acid. Often used in industrial settings and laboratories, sulfuric acid is an oxidizing agent that enhances the combustion of other properties, and is ultimately what helps energize batteries.
How do you make H2SO4 solution?
If you took 6.9 mL of concentrated sulfuric acid and diluted it to 250 mL, you would have a 1 N H2SO4 solution. (Important note: Always add the acid (or base) to water, in that order. Pour slowly with constant mixing.
What is the common name of Sulphuric acid?
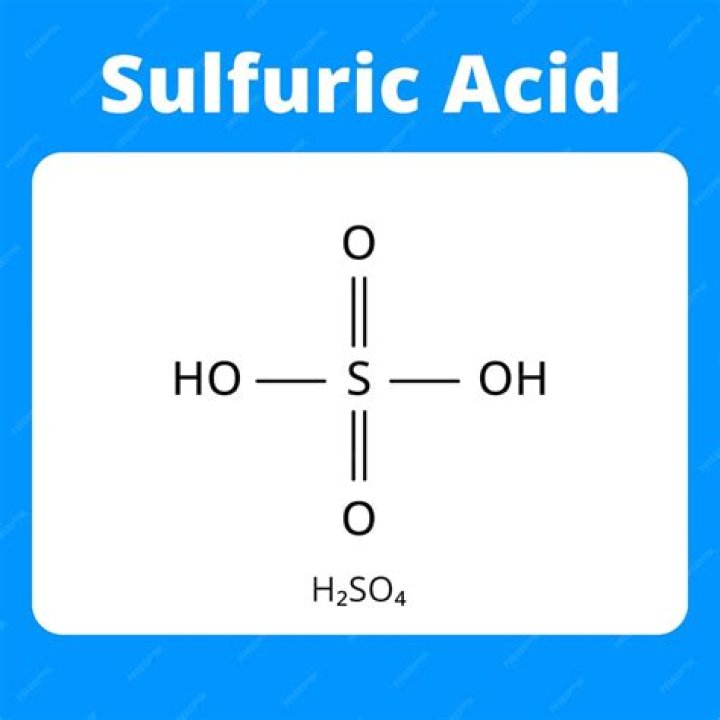
sulfuric acid, sulfuric also spelled sulphuric (H2SO4), also called oil of vitriol, or hydrogen sulfate, dense, colourless, oily, corrosive liquid; one of the most commercially important of all chemicals.
What is the acid used in car batteries?
The electrolyte is sulfuric acid (H2SO4) in water.
Is sulfuric acid used in pharmaceuticals?
Sulfuric acid in the production of drugs: Sulfuric acid can damage the cancerous cell DNA, hence used to manufacture chemotherapy drugs.
How do you make sulfur from sulfuric acid?
Take the sulfur and burn it with oxygen to make SO2. Then, direct the stream of SO2 gas into a reactor bed with some vanadium pentoxide catalyst and extra oxygen gas to make sulfur trioxide gas which is the acid anhydride of sulfuric acid. Then, the bubble the SO3 gas produced by that reaction into some water.
Where can you find natural sulfur?
It is the fifth most common element by mass in the Earth. Elemental sulfur can be found near hot springs and volcanic regions in many parts of the world, especially along the Pacific Ring of Fire; such volcanic deposits are currently mined in Indonesia, Chile, and Japan.
What is the liquid inside a battery?
The battery contains a liquid electrolyte such as sulfuric acid, a dangerous corrosive liquid.
What liquid is in a battery?
Battery fluid, a mixture of sulfuric acid and distilled water (called electrolyte), creates the electricity that makes a modern battery work so efficiently. Depending on the type of battery in your vehicle, battery fluid can evaporate and over time will need to be topped off as part of regular battery care.
Can I use distilled water instead of battery acid?
The electrolyte in a flooded deep cycle battery is a solution of acid and water. Using distilled or deionized water is recommend because it is free from additional minerals you would find in water such as regular tap water. … Never add additional acid or electrolyte solution, this can also diminish the battery life.
Can you pour sulfuric acid down toilet?
You can clean out a blockage in your toilet drain with sulfuric acid. … Cleaning out a toilet drain with a chemical substance, such as sulfuric acid, can often unclog the blockage and restore function to your toilet. However, you’ll need to proceed with caution, as sulfuric acid is an extremely toxic substance.
What happens when you mix sulfuric acid and Drano?
However, drain cleaners have some extremely toxic ingredients, and mixing two brands of drain cleaner can create a poisonous situation or even cause an explosion. … For example, if you mix a drain cleaner with sulfuric acid and one with sodium hydroxide, you get an acid-base reaction, resulting in salt and water.
Do batteries contain acid?
Batteries contain acid because it’s fundamental to the electrochemical reaction that takes place. Also referred to as battery electrolyte, battery acid is the medium that carries the electrical flow between positive and negative electrodes.
Why is there sulfuric acid in a car battery?
When you hear about electrolyte in reference to car batteries, what people are talking about is a solution of water and sulfuric acid. This solution fills the cells in traditional lead acid car batteries, and the interaction between the electrolyte and the lead plates allows the battery to store and release energy.
Why do car batteries use sulfuric acid?
The formation of the lead sulphate products involves sulphuric acid, which is the electrolyte used in these batteries. … Because sulphuric acid is consumed during the discharge process and released during the charge process, the specific gravity of the electrolyte changes during battery use and charge.
How do you make 2 normal H2SO4?
Preparation of H2SO4 – 2 Molar (4 Normal, 4N) = 11.1 mL of concentrated H2SO4 to 100 mL deionized water. 1 Molar (2 Normal, 2N) = 5.5 mL concentrated H2SO4 to 100 mL deionized water. 0.5 Molar (1 Normal, 1N) = 2.7 mL concentrated H2SO4 to 100 mL deionized water.
How do you make 1 mole of H2SO4?
- The Molecular weight of is 98 g/mol.
- Next we add 98g of conc. …
- As Acid is a liquid,density should be taken into account.
- Density =Mass/volume.
- Density given on the bottle of Sulphuric acid is 1.84 g/ml.
How do you make 0.1 H2SO4?
Preparation of 0.1 N Sulphuric acid (H2SO4) Solution Take 3.0 mL of concentrated Sulphuric acid (H2so4, sp gr 1.84) into a 1000-ml volumetric flask. Dilute to the mark with water, mix well, and store in a tightly closed glass container.
Why is Sulphuric acid called King of chemicals?
Sulphuric acid is called “king of acids” because of its direct and indirect applications in the manufacture of many chemicals including fertilizers. Sulphuric acid is used to clean up rust from steel rolls and soap. It also dissolves its compounds. Thus, it is ideal to call sulphuric acid as king of chemicals.
Is hydrogen sulfate the same as sulfuric acid?
The HSO4- ion is commonly referred to as hydrogen sulfate (which is also called the bisulfate ion). H2SO4 can be called dihydrogen sulfate (but it is popularly known as sulfuric acid).
How is sulfuric acid made in industry?
Manufacture. Sulfuric acid is produced from sulfur, oxygen and water via the contact process. In the first step, sulfur is burned to produce sulfur dioxide. This is then oxidised to sulfur trioxide using oxygen in the presence of a vanadium(V) oxide catalyst.