What Subshell has 3 orbitals
A p subshell has three orbitals. A d subshell has five orbitals.
How does P subshell have 3 orbitals?
Since electrons all have the same charge, they stay as far away as possible because of repulsion. So, if there are open orbitals in the same energy level, the electrons will fill each orbital singly before filling the orbital with two electrons. For example, the 2p shell has three p orbitals.
Is subshell 3s possible?
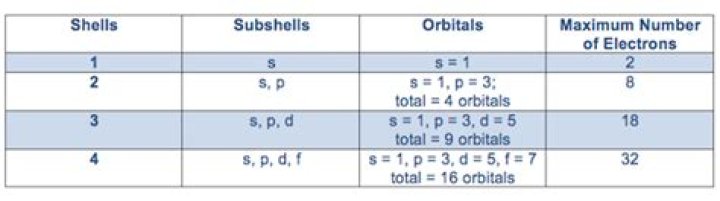
ShellSubshellTotal Number of Electrons in Shell3rd Shell3s, 3p, 3d2 + 6 + 10 = 184th Shell4s, 4p, 4d, 4f2 + 6 + 10 + 14 = 32
Does every P subshell have 3 orbitals?
There are three atomic orbitals in a p subshell. … When each orbital (x,y,z) is full with 2 electrons, then the p subshell holds a total of 6 electrons.Does 4p have 3 orbitals?
For any atom, there are three 4p orbitals. These orbitals have the same shape but are aligned differently in space. The three 4p orbitals normally used are labelled 4px, 4py, and 4pz since the functions are “aligned” along the x, y, and z axes respectively. Each 4p orbital has six lobes.
How many orbitals are allowed in a subshell of L 3?
How many orbitals are allowed in a subshell if the angular momentum quantum number for electrons in that subshell is 3? Also, l=3 is an F orbital which has 14 electrons and 7 orbitals.
How many orbitals are in the N 3?
There are nine orbitals in the n = 3 shell. There is one orbital in the 3s subshell and three orbitals in the 3p subshell. The n = 3 shell, however, also includes 3d orbitals. The five different orientations of orbitals in the 3d subshell are shown in the figure below.
How many orbitals does f subshell have?
So, mlvalues are +3,+2,+1,0,−1,−2,−3 which makes total of 7 orbitals of f subshell.How many orbitals does d subshell have?
electronic configuration called p orbitals; and a d subshell (l = 2) consists of five orbitals, called d orbitals.
Which of the following orbitals has three angular nodes?4f has 3 angular nodes, 7p has 1 angular node, 6d has 2 angular nodes and 3p has 1 angular node.
Article first time published onWhat sub shells are possible in n 3 energy level how many orbitals of all kinds are possible in this level?
What subshells are possible in n = 3 energy level? For n = 3 energy level, the possible values of l are 0, 1 and 2. Thus, there are three subshells namely: l = 0, s subshell ; l = 1, p subshell ; and l = 2, d subshell.
How many electrons can n 3 hold?
2 electrons are the maximum number of electrons that can exist in any orbital. In the n = 3 shell, 2 electrons max fit into the s subshell and 6 electrons max in the 3px, 3py, and 3pz. The maximum number of electrons that can fit inside a ‘n’ shell is 2n^2. So it is 18.
How many orbitals have n 3 and ML 1 Enter your answer as an integer without units?
Each ml value represents one orbital. So, there are two orbitals that have n = 3 and ml=−1 m l = − 1 .
How many orbitals are in the 5d sublevel?
Therefore, the 3d-subshell will contain a total of five 3d-orbitals. Likewise, the 4d-subshell will contain a total of five 4d-orbitals, the 5d-subshell will contain a total of five 5d-orbitals, and so on.
Where is 3d orbital?
The 3d orbitals are quite compactly arranged around the nucleus. Introducing a second electron into a 3d orbital produces more repulsion than if the next electron went into the 4s orbital. There is not a very big gap between the energies of the 3d and 4s orbitals.
How many orbitals are in the 4d subshell?
Now we can say that $4d$ has five orbitals having magnetic quantum numbers from $( – 2)\,\,to\,( + 2)$ .
How many quantum states are there in n 3?
distinct orbitals. So, if each electron is described by an unique set of quantum numbers, you can conclude that 18 sets of quantum numbers are possible for the third energy level.
How many orbitals are possible in 5g subshell?
There are nine different orbitals in the 5g subshell. As the orbitals proceed sequentially from s to f, two additional orbitals are added to each…
Which azimuthal quantum numbers can exist for n 3?
For example, if n =3, the azimuthal quantum number can take on the following values – 0,1, and 2. When l=0, the resulting subshell is an ‘s’ subshell.
How many orbitals are in 3rd principal energy level?
So, the third principal energy level can hold maximum 18 electrons and contain one s-orbital, three p-orbital and five d-orbital. Therefore, the third main energy level contains one s-orbital, three p-orbital and five d-orbital so the total orbitals will be 9.
How many orbitals does N 3 L 3 have?
The 3s subshell will be having only 1 orbital. The 3p subshell will be having 3 orbitals. The 3d subshell will be having 5 orbitals. On adding all the orbitals of 3s, 3p and 3d we get a total of 9 orbitals.
How many orbitals does s subshell have?
Thus the s subshell has only one orbital, the p subshell has three orbitals, and so on.
Which orbitals are electrons with L 3 in?
That is a d orbital. So for l = 3, the wave function will have three nodes. Those are the f orbitals.
What is the subshell notation for quantum numbers n 3 and L 2?
This is a 3d orbital, because n =3 and l = 2 which is a d-subshell. Thus, this orbital is of the 3rd shell, and the d-subshell.
How many orbitals are in P block?
The p sublevel has 3 orbitals, so can contain 6 electrons max. The d sublevel has 5 orbitals, so can contain 10 electrons max. And the 4 sublevel has 7 orbitals, so can contain 14 electrons max.
How many orbitals are in the 4f sublevel?
For any atom, there are seven 4f orbitals. The f-orbitals are unusual in that there are two sets of orbitals in common use.
How many orbitals are in the 4f subshell?
As such, for a 4f subshell, the number of orbitals is 7.
What can be the representation of the orbital having 3?
And total nodes is n-1 which is equal to 3. which implies n=4. l=3 represents the orbital d orbital and n=4 shows that its in 4th shell. therefore the required orbital is 4d.
How many electrons can fit in the orbital for which N 3 and i 1?
So, n=3 and l = 1 indicates electrons are present in the 3p subshell. The p subshell has a maximum of 3 orbitals and each orbital contains a maximum of 2 electrons. Hence, the correct option is (D) 6.
What is angular momentum of an electron in 4f orbital?
Angular momentum of the electron in the 4f-orbital of a one-electron species according to wave mechanics is- √12hπ.
How many Subshells are there in 3rd shell?
The third shell has three subshells, labeled s, p, and d. The d subshell can hold a maximum of 10 electrons.



