What theory explains the behavior of gases
The Kinetic-Molecular Theory Explains the Behavior of Gases, Part II. According to Graham’s law, the molecules of a gas are in rapid motion and the molecules themselves are small. The average distance between the molecules of a gas is large compared to the size of the molecules.
What theory is used to explain the behavior of particles in matter?
The kinetic theory of matter (particle theory) says that all matter consists of many, very small particles which are constantly moving or in a continual state of motion.
How does the particle theory explain the properties of gases?
The kinetic particle theory explains the properties of the different states of matter. The particles in solids, liquids and gases have different amounts of energy. They are arranged differently and move in different ways.
What is the theory of ideal gases?
An ideal gas is a gas where the atoms do not exert forces on each other but they do collide with the walls of the container (in elastic collisions).What is wave theory and particle theory?
The theory is often called the wave-particle theory. It explains how electromagnetic radiation can behave as both a wave and a particle. … The theory posits that waves of photons traveling through space or matter make up electromagnetic radiation.
What are postulates of kinetic theory of gases?
The five main postulates of the KMT are as follows: (1) the particles in a gas are in constant, random motion, (2) the combined volume of the particles is negligible, (3) the particles exert no forces on one another, (4) any collisions between the particles are completely elastic, and (5) the average kinetic energy of …
What are the kinetic theory assumptions about ideal gases?
The simplest kinetic model is based on the assumptions that: (1) the gas is composed of a large number of identical molecules moving in random directions, separated by distances that are large compared with their size; (2) the molecules undergo perfectly elastic collisions (no energy loss) with each other and with the …
Why is the kinetic theory of gases important?
The kinetic theory of gases is important for clarifying the capture process of particles by the diffusion mechanism. According to this theory, gas is composed of a large number of small-sized molecules compared with the distances between them.What is the kinetic theory in chemistry?
Kinetic Molecular Theory states that gas particles are in constant motion and exhibit perfectly elastic collisions. … The average kinetic energy of a collection of gas particles is directly proportional to absolute temperature only.
What is kinetic theory GCSE?The kinetic theory model can be used to explain the properties of solids, liquids and gases by considering the molecules they are made from and their motion. For gases, the kinetic theory model explains that gas pressure is caused by the collisions between the particles and their container.
Article first time published onHow particle Behaviour is influenced by the energy of the particles?
Particles move rapidly in all directions but collide with each other more frequently than in gases due to shorter distances between particles. With an increase in temperature, the particles move faster as they gain kinetic energy, resulting in increased collision rates and an increased rate of diffusion.
What is wave theory?
Definition of wave theory : a theory in physics: light is transmitted from luminous bodies to the eye and other objects by an undulatory movement.
How does the corpuscle theory explain light's nature?
The corpuscular theory explains the reflection of light in exactly the same way as the reflection of a perfectly elastic ball from a rigid plane. When the corpuscles (particles) hit the reflecting surface, they are reflected from it in such a way that the angle of incidence is equal to the angle of reflection.
What is difference between wave theory and particle theory of light?
The key difference between wave and particle nature of light is that the wave nature of light states that light can behave as an electromagnetic wave, whereas the particle nature of light states that light consists of particles called photons. Wave-particle duality is a concept in quantum mechanics.
What is kinetic theory of gases Class 11?
Kinetic molecular theory of gases. Kinetic molecular theory of gases. Molecules are point masses having no volume. Gas atoms apply no constrain on different atoms unless they suffer collision. Collisions of particles with each other or with the boundaries of container do not result in decreased energy system.
How does the behaviour of real gas is tested for ideal behaviour?
Real gases exhibit ideal behaviour only when the intermolecular forces are minimal. The lesser the pressure, the greater the chances of a real gas behaving like an ideal gas! Let Z = pV/nRT be a number. it will have no units as is clear from the equation.
What does the kinetic theory of gases say about gases in general?
The model, called the kinetic theory of gases, assumes that the molecules are very small relative to the distance between molecules. The molecules are in constant, random motion and frequently collide with each other and with the walls of any container. … The higher the temperature, the greater the motion.
Why is kinetic theory explained?
Kinetic theory explains the behaviour of gases based on the idea that the gas consists of rapidly moving atoms or molecules. … It gives a molecular interpretation of pressure and temperature of a gas, and is consistent with gas laws and Avogadro’s hypothesis. It correctly explains specific heat capacities of many gases.
What is kinetic energy theory?
Kinetic energy is energy that an object has because of its motion. The Kinetic Molecular Theory explains the forces between molecules and the energy that they possess. … Temperature is the term used to explain how hot or cold an object is. Temperature is the average kinetic energy of particles in the substance.
How the kinetic theory explains the expansion of metals liquids and gases?
Metal railway tracks have small gaps so that when the sun heats them, the tracks expand into these gaps and don’t buckle. … Heat causes the molecules to move faster, (heat energy is converted to kinetic energy ) which means that the volume of a gas increases more than the volume of a solid or liquid.
What is the particle theory Grade 7?
The particle theory of matter is: A scientific model of the structure of matter; according to the particle theory, all matter is made up of extremely tiny particles, and each pure substance has its own kind of particle, different from the particles from any other pure substance.
What is a theory that explains the properties of solid liquids and gases by proposing rapidly moving tiny particles either atoms or molecules or three words?
After issuing a fiery _____ questioning the foreign king’s intentions, Queen Alana began lo prepare for war. Two days later, the _____, old-world forest on the southern shore of her realm became crowded with invading troops, all of whom were preparing for an onslaught on the supposedly _____ castle.
How gases behave based on the motion and relative distances between gas particles?
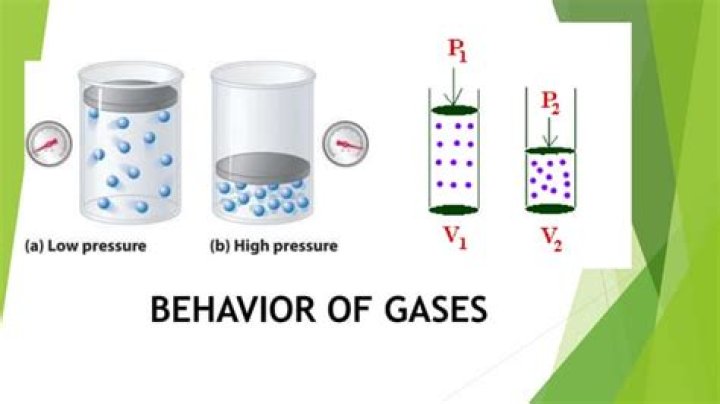
If a given gas sample is allowed to occupy a larger volume, then the speed of the molecules does not change, but the density of the gas (number of particles per unit volume) decreases, and the average distance between the molecules increases. Hence the molecules must, on average, travel farther between collisions.
Which statement describes a limitation of the kinetic-molecular theory for a gas?
Which statement describes a limitation of the kinetic-molecular theory for a gas? The theory assumes that particles do not experience intermolecular forces. The theory states that pressure is inversely proportional to volume.
How do you use Elliott wave theory?
To use the theory in everyday trading, a trader might identify an upward-trending impulse wave, go long and then sell or short the position as the pattern completes five waves and a reversal is imminent.
Does the Elliott wave theory work?
Yes Elliott wave works and it works amazingly as well but it can get complicated. The market for some reason moves in a specific way, I can’t explain why but its like natures laws also governs the market.
What is corpuscular theory of Newton?
Isaac Newton argued that the geometric nature of reflection and refraction of light could only be explained if light were made of particles, referred to as corpuscles because waves do not tend to travel in straight lines. Newton sought to disprove Christiaan Huygens’ theory that light was made of waves.
What is Huygens wave theory principle?
Huygens’ principle, in optics, a statement that all points of a wave front of light in a vacuum or transparent medium may be regarded as new sources of wavelets that expand in every direction at a rate depending on their velocities.
What is Huygens wave theory of light?
In 1678, Huygens proposed that every point that a luminous disturbance meets turns into a source of the spherical wave itself. The sum of the secondary waves, which are the result of the disturbance, determines what form the new wave will take. This theory of light is known as the ‘Huygens’ Principle’.
Who proposed the wave theory of light?
Wave theory of light was proposed by Huygens in 1678. Wave theory proposed by Huygens: Light travels in a medium in a form called a wavefront.
Which theory of light is the photon more consistent with the wave theory or the particle theory?
Which theory of light is photon more consistent w/, the wave or particle theory? The photon is considered to be a light particle, which plays more in the theory that light is actually a particle.



