What type of compound is nf3
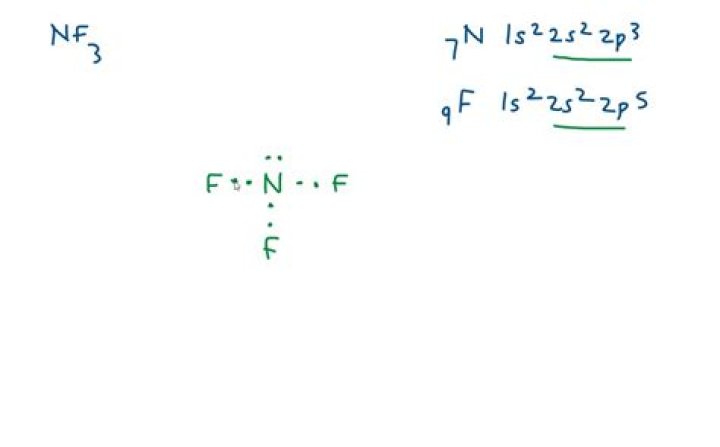
Nitrogen trifluoride | NF3 – PubChem.
Is NF3 an ionic or covalent compound?
So, the difference in electronegativity between N and F is (4.0-3.0) or 1.0. As the electronegativity difference is less than 1.8, therefore each bond between N and F is covalent. Hence, NF3 N F 3 is a molecular compound.
Is NF3 covalent in nature?
The molecule BF3 and NF3 , both are covalent compounds but BF3 is non – polar and NF3 is polar.
Is NF3 a nonpolar covalent bond?
NF3 is polar. The reason for this is because in NF3 there is a pair of electrons on Nitrogen which are not taking part in bonding, making it polar.How many covalent bonds are in NF3?
NF 3 (Nitrogen trifluoride) Lewis Structure | Steps of Drawing. In the lewis structure of Nitrogen trifluoride (NF3), there are three N-F bonds and one lone pair on nitrogen atom. Each fluorine atom has three lone pairs.
What is the shape of NF3?
The molecule has a trigonal pyramidal structure, like ammonia.
What is dipole moment NF3?
Answer: N- atom is the central atom of NF3 and NH3. … As, F-atom is more electronegativity than H- atom,NF3 should have higher dipole moment than NH3. The dipole moment of NH3 = 1.46D. The dipole moment of NF3 = 0.24D.
Is NF3 is more covalent than bf3?
NF3 and BF3 both are covalent but BF3 is non polar , while NF3 is polar because there is no dipole moment in BF bond while NF bond have some dipole moment.Is NF3 more covalent than BiF3?
BIn NF3 N and F are non-metals but BiF3 Bi is metal but F is non-metal therefore NF3 is more covalent than BiF3.
Is CH3OH a hydrocarbon?Methanol is a simplest alcohol with a chemical formula CH3OH. It is not a hydrocarbon since the hydroxyl group is chemically bonded to the carbon atom. It consists of a methyl group linked with a hydroxy group. It is also known as Wood alcohol or Methyl alcohol.
Article first time published onWhat kind of bond is CH3OH?
The bond between oxygen and hydrogen in the O-H is polar covalent because the electronegativity of oxygen is 3.44, while the EN for hydrogen is 2.20,…
What kind of solid is CH3OH?
It is an alkyl alcohol, a one-carbon compound, a volatile organic compound and a primary alcohol. It is a conjugate acid of a methoxide. Methanol is released to the environment during industrial uses and naturally from volcanic gases, vegetation, and microbes.
Is NF3 dipole dipole?
There is one lone pair around the central nitrogen atom, along with three bonds. So, the VSEPR shape of NF3 will be trigonal pyramidal, which means that it’s a polar molecule. … The polarity of NF3 causes there to not only be London dispersion forces (which are present in every molecule), but also dipole-dipole forces.
Is NF3 a stable molecule?
NF3 is stable as the sizes of fluorine and nitrogen are comparable and the bonding interaction is good enough to give a stable compound.
Which is correct for dipole moment NH3 and NF3 respectively?
Dipole moment of NF3 is less than that of NH3.
What is the structure of NF3 according to Vsepr theory?
According to the VSEPR chart, the molecular geometry of nitrogen trifluoride is trigonal bipyramidal. We can also predict electron geometry via electron groups through VSEPR theory. In NF3, the central nitrogen atom has four electron groups surrounding it: three single bonds( three bonded pairs) and one lone pair.
Is BiF3 ionic or covalent?
Bismuth is a metal and fluorine is a non-metal. Also, the electronegativity of fluorine is the highest and hence, greater electronegativity difference between them, makes BiF3 ionic.
Why is NF3 stable?
– F has very high electronegativity as compared to Cl & I. – As electronegativity difference between N & F is very big it forms a strong N-F bond and thus stable NF3 molecule.
Why BiF3 is covalent in nature?
Bi is the higher member of Nitrogen group. As we move down the group , the tendency of donating electrons increases. This leads to the increasing of ionic character of atom that favours the ionic bond. That is why BiF is ionic not covalent.
Which of the following compounds are non polar NF3?
The unsymmetrical geometry of $N{F_3}$ molecule causes a non-zero dipole moment in the molecule. The molecule $B{F_3}$ and $N{F_3}$ both are covalent compounds but $B{F_3}$ is non-polar and $N{F_3}$ is polar, the reason is that $B{F_3}$ is symmetrical molecule whereas $N{F_3}$ is unsymmetrical.
Which has regular geometry NF3 or BF3?
Shape of NF3 is pyramidal while the shape of BF3 is triangular planar.
Is alcl3 ionic or covalent?
2 Answers. Ernest Z. The Al-Cl bond in AlCl₃ is not ionic — it is polar covalent. The electronegativity of Al is 1.5.
What is CH3OH in chemistry?
Methanol (CH3OH), CAS 67-56-1, also known as methyl alcohol or MeOH, is the simplest alcohol. Historically known as wood alcohol, methanol is a light, colorless, highly flammable, and volatile liquid.
Is CH3OH an electrolyte?
Methanol, CH3OH, is a nonelectrolyte; ammonia, NH3, is a weak electrolyte; and iron(III) sulfate, Fe2(SO4)3, is a strong electrolyte.
What type of compound is methanol CH3OH?
methanol (CH3OH), also called methyl alcohol, wood alcohol, or wood spirit, the simplest of a long series of organic compounds called alcohols, consisting of a methyl group (CH3) linked with a hydroxy group (OH).
Is there hydrogen bonding in CH3OH?
Methanol exhibits hydrogen bonding, and all three of the van der Waals attractions: Keesom forces (dipole-dipole) attraction, Debye forces (induced attraction) and London dispersion forces (which all molecules exhibit). Methanol is polar and has a net dipole moment of 1.69 Debye.
What predominant intermolecular force is in CH3OH?
Methanol (CH3OH C H 3 O H ) The predominant intermolecular force in methanol is hydrogen bonding.
What are molecular solids examples?
Molecular solids have low melting (Tm) and boiling (Tb) points compared to metal (iron), ionic (sodium chloride), and covalent solids (diamond). Examples of molecular solids with low melting and boiling temperatures include argon, water, naphthalene, nicotine, and caffeine (see table below).
What type of solid is I2?
Iodine (I2) consists of larger, nonpolar molecules and forms a molecular solid that melts at 114 °C.



