What type of reaction takes place when atoms or molecules rearrange to form new substances
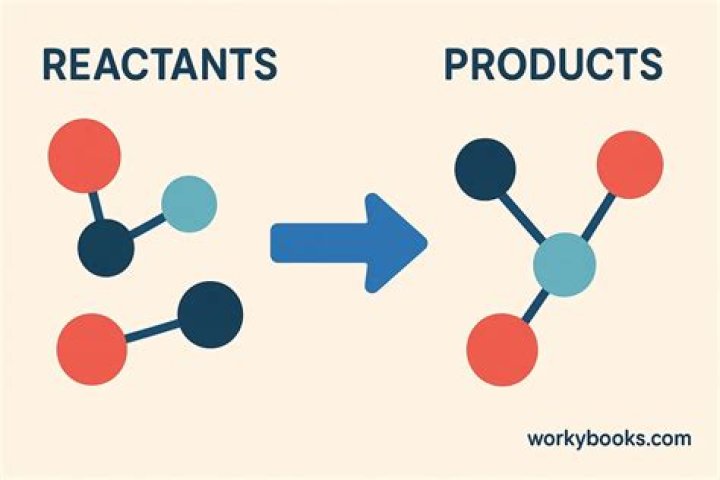
In a chemical reaction, bonds between atoms in the reactants are broken and the atoms rearrange and form new bonds to make the products.
What two substances are produced when atoms rearrange?
A chemical reaction is the process in which atoms present in the starting substances rearrange to give new chemical combinations present in the substances formed by the reaction. These starting substances of a chemical reaction are called the reactants, and the new substances that result are called the products.
Do atoms rearrange during a chemical reaction?
During a chemical reaction no atoms are created or destroyed. The atoms are rearranged. This results in the formation of new substances with different properties to the starting substances.
What molecules combine to form new substances?
When two or more elements combine to form a new substance, it is called a compound. There are many different types of compounds, because atoms of elements combine in many different ways to form different compounds. Examples include water (H2O) and table salt (NaCl).Are the new substances that are formed?
A change in which one or more new substances are formed is called a chemical change. A chemical change is also called a chemical reaction. Chemical changes are very important in our lives. All new substances are formed as a result of chemical changes.
How does a chemical reaction form new substances?
In a chemical change, new substances are formed. In order for this to occur, the chemical bonds of the substances break, and the atoms that compose them separate and rearrange themselves into new substances with new chemical bonds. When this process occurs, we call it a chemical reaction.
How do atoms combine to form new substances?
Chemical reactions happen when chemical bonds are broken and formed. A chemical bond is a force that holds two atoms together in a molecule. During a chemi- cal reaction, some of the bonds in the original molecule break. New bonds form to produce a new substance.
How do substances change into different substances during chemical reactions?
Chemical reactions change substances into different substances by breaking and forming chemical bonds. The oxygen molecules (O2) that you breathe in are part of a series of chemical reactions.How atoms are held together to form substances?
The atoms in chemical compounds are held together by attractive electrostatic interactions known as chemical bonds. Most covalent compounds consist of molecules, groups of atoms in which one or more pairs of electrons are shared by at least two atoms to form a covalent bond.
What changes occur during chemical reaction?Chemical reactions involve combining different substances. The chemical reaction produces a new substance with new and different physical and chemical properties. … The particles of one substance are rearranged to form a new substance. The same number of particles that exist before the reaction exist after the reaction.
Article first time published onWhy do atoms take part in chemical reactions?
Atom take parts in chemical reaction in order to gain it’s stable electronic configuration i.e Noble gas electronic configuration.
Are the starting substances in the reaction while are the new substances that are formed?
Reactants are the starting substances in the reaction while products are the new substances that are formed.
How atoms unite and change into molecules?
When atoms join together to form molecules, they are held together by chemical bonds. These bonds form as a result of the sharing or exchange of electrons between the atoms. … Different atoms use these electrons to form one of three different types of bond: ionic bonds, covalent bonds, or metallic bonds.
What happens when atoms collide?
Our existence, as well as our ability to touch and interact with the universe around us, is governed by the collisions of atoms and molecules. The constant jostling and vibrating of these particles give us heat, light, and life.
How are atoms rearranged during a combustion reaction?
When things burn, it looks like they are destroyed, but during any chemical reaction no particles are created or destroyed. The atoms in fuels are simply rearranged from the reactants to the products during combustion. The products may have different properties to the reactants.
What are the 4 types of chemical reactions?
Four basic types Representation of four basic chemical reactions types: synthesis, decomposition, single replacement and double replacement.
What are reaction types?
The five basic types of chemical reactions are combination, decomposition, single-replacement, double-replacement, and combustion. Analyzing the reactants and products of a given reaction will allow you to place it into one of these categories. Some reactions will fit into more than one category.
Is molecules take part in chemical reaction?
Yes, molecules take part in the chemical reaction. A group of atoms bonded together are called molecules. Molecules interact with each other as reactants in a chemical reaction and produce new molecules as products.
How do atoms take part in a chemical reaction?
During a chemical reaction, the atoms of the original substances gain, lose or share their electrons with those of the substances with which they are reacting. The reaction creates new substances made up of a new combination of atoms and a different configuration of electrons.
What are two types of common reactions?
The more common types of chemical reactions are as follows: Combination. Decomposition. Single displacement.
What is the name given for the substance that react and form in a chemical reaction?
The substance which reacts in a chemical reaction is called as REACTANT and the substance form is called as PRODUCT.
Which substances form due to a chemical reaction?
Answer: Substances that are formed as a result of a chemical reaction are called products and are written on the right-hand side of the equation.
Why do atoms bond to form molecules?
Atoms come together to form molecules because of their electrons. … When two atoms share electrons between them, they are locked together (bonded) by that sharing. These are called covalent bonds. Bonds like this are in oxygen gas, nitrogen gas, and hydrogen gas.
Why do atoms combine together and form molecules?
Atoms combine to form a molecule in order to achieve a stable configuration like those of the noble gases.
Why do atoms take part in bond formation?
Atoms form chemical bonds to make their outer electron shells more stable. The type of chemical bond maximizes the stability of the atoms that form it. … Covalent bonds form when sharing atoms results in the highest stability. Other types of bonds besides ionic and covalent chemical bonds exist, too.
What is it called when two atoms collide?
The collide . . . they rebound. Done. Just two atoms do not have a capability of absorbing the energy of impact. If this were to happen when one of the atoms was attached to a surface, then a reaction might occur.