Which is the anomeric carbon in fructose
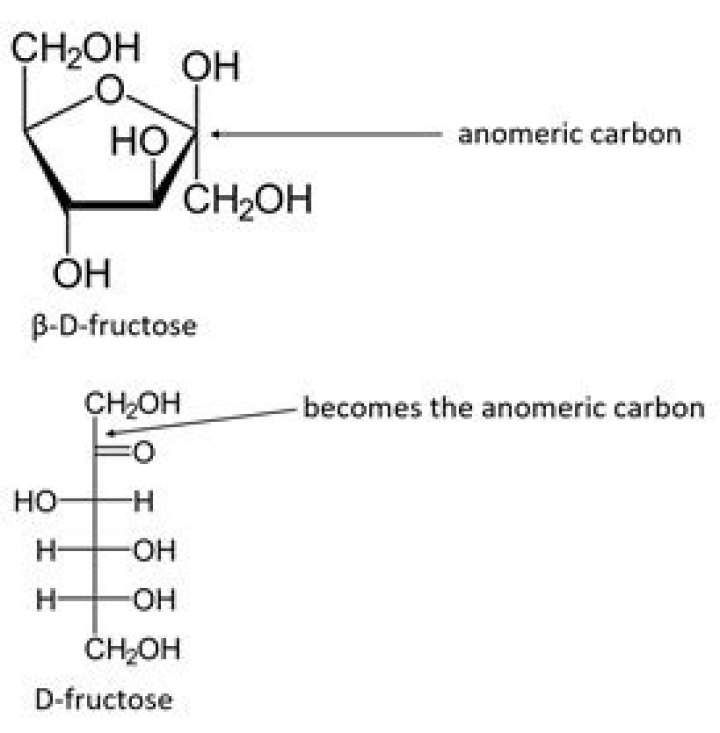
In D-fructose, the carbonyl group is at C-2 . Here, C-2 is the anomeric carbon.
Does fructose have two anomeric carbons?
In fructose, both anomeric carbons are in acetal functional groups, so fructose is a non-reducing sugar. This brings us to the topic of disaccharides. The linkages between the monosaccharide ring units in disaccharides are acetal linkages.
How do you find the anomeric carbon?
In the cyclic form, the anomeric carbon can be found next to the oxygen atom in the pyranose or furanose ring, but on the opposite side from the carbon that carries the acyclic CH2O group (e.g., the CH2OH group in the example shown here).
Which is the anomeric carbon?
The anomeric carbon is the carbon derived from the carbonyl carbon compound (the ketone or aldehyde functional group) of the open-chain form of the carbohydrate molecule. Anomerization is the process of conversion of one anomer to the other.What is anomeric carbon example?
An example of anomeric carbon is that carbon in a monosaccharide (like glucose) about which rotation occurs. The anomeric carbon can be determined by the carbon (C) attached to two oxygen (O) atoms joined by single bonds. This rotation brings about two distinct configurations, α and β -anomers.
Is fructose a 5 carbon sugar?
Fructose is recognized by having a five member ring and having six carbons, a hexose. While fructose is a hexose, it typically exists as a 5-membered hemiacetal ring (6 carbon sugar), The aldoses are glucose and galactose, and the ketose is fructose. …
How many carbons are in fructose?
Fructose is another sugar that also has 6 carbons, 12 hydrogens, and 6 oxygen atoms. However, the arrangement of the atoms is different, and this makes it much sweeter than glucose and also affects its ability to combine with other molecules.
What are anomers 12 chemistry?
Anomers are cyclic monosaccharides, differing from each other in the configuration of C-1 carbon or C-2 carbon. For aldoses, it is C-1 and C-2 for ketoses. The distinguishing carbon atom is called anomeric carbon or anomeric center. We will now draw the pair of anomers for the monosaccharide, glucose.Which are the anomers?
Anomers are cyclic monosaccharides or glycosides that are epimers, differing from each other in the configuration of C-1 if they are aldoses or in the configuration at C-2 if they are ketoses. Example 1: α-D-Glucopyranose and β-D-glucopyranose are anomers. …
When fructose adopts a Furanose structure which carbon is the anomeric carbon?The C-1 carbon is called the “anomeric” carbon and the α and β diastereoisomers are referred to as “anomers”.
Article first time published onHow do you count carbons in sugar?
Carbon atoms are numbered beginning from the reactive end of the molecule, the CHO (aldehyde) or “C” double bonded “O” (carbonyl) end of the molecule. Each carbon atom is then numbered in order through the end of the chain.
What is anomeric carbon in lactose?
A common disaccharide is lactose, which is found only in milk. … Lactose is formed from monosaccharides β -D- galactose and β -D- glucose . The anomeric carbon of the β -D-galactose molecule reacts with the C-4 hydroxyl group of the β -D-glucose molecule to form the glycosidic bond (see Figure 1).
Where are the anomeric carbons in lactose?
Carbon # 1 (red on left) is called the anomeric carbon and is the center of an acetal functional group. A carbon that has two ether oxygens attached is an acetal. The Beta position is defined as the ether oxygen being on the same side of the ring as the C # 6.
What is the anomeric carbon in sucrose?
During the formation of sucrose, both the glucose and fructose molecules’ anomeric carbons get involved to form the O-glycosidic bond. An anomeric carbon is the carbon that was once a part of the carbonyl group of the sugar molecule before it reacts internally with its own hydroxyl group.
Which sugars have a free anomeric carbon?
Contrarily, maltose and lactose, which are the reducing sugar, have a free anomeric carbon that can get converted into an open-chain form by forming a bond with the aldehyde group. Figure 1: Chemical Structure of Glucose, Fructose, and Sucrose.
How many anomeric carbons are in lactose?
Reducing disaccharides like lactose and maltose have only one of their two anomeric carbons involved in the glycosidic bond, while the other is free and can convert to an open-chain form with an aldehyde group.
How many chiral carbons are in fructose?
Hence, the number of chiral atoms in glucose and fructose are 4 each.
Is fructose a furanose?
In solution, glucose is mostly in the pyranose form, fructose is 67% pyranose and 33% furanose, and ribose is 75% furanose and 25% pyranose. Sugars can be drawn in the straight chain form as either Fisher projections or perspective structural formulas.
Is fructose 5 carbon or 6 carbon sugar?
Glyceraldehyde and dihydroxyacetone are trioses (3-carbon atoms), ribose is a pentose (5-carbon atoms), while glucose, fructose, and galactose are hexoses (6-carbon atoms) (Fig.
Why is fructose a furanose?
Fructose also exists in two cyclic forms which are obtained by the addition of —OH at C5 to the C = O group. The ring, thus formed is a five membered ring and is named as furanose with analogy to the compound furan.
What type of compound is fructose?
Fructose is a carbohydrate, an organic compound in which five of the six carbon atoms are arranged in a ring to which are attached the hydrogen atoms and hydroxy (−OH) groups that make up the molecule.
What are anomers 11?
Anomers are cyclic monosaccharides or glycosides that are epimers, differing from each other in the configuration of C-1 if they are aldoses or in the configuration at C-2 if they are ketoses. … Alpha and beta anomers are formed from an open-chain sugar by mutarotation.
Are glucose and fructose anomers?
An anomer is a distinct type of epimer in which one of two stereoisomers of a cyclic saccharide differs only in its configuration at the acetal carbon, also called the anomeric carbon. Glucose and fructose are not anomers.
What is alpha and beta anomers?
The key difference between alpha and beta anomers is that in alpha anomer the hydroxyl group at the anomeric carbon is cis to the exocyclic oxygen at the anomeric centre, whereas in beta anomer the hydroxyl group is trans to the exocyclic oxygen. … It is a type of epimer in which we can see an anomeric carbon atom.
What are anomers Byjus?
Anomers are cyclic monosaccharides or glycosides that are epimers, differing from each other in the configuration of C-1 if they are aldoses or in the configuration at C-2 if they are ketoses. The epimeric carbon in anomers are known as anomeric carbon or anomeric centre.
What are anomers Toppr?
Two compounds with more than one chiral carbon that deffer from one another at one carbon atom are called as anomers.
What are anomers give two anomers of glucose?
C-1 of glucose become chrial carbon and has two possible arrangements of H and OH group around itThese two arrangements of glucose which differ only in the orientation of hydroxyl group at C-1 are called anomers. Eg:– α-D-Glucose and β-D-Glucose.
What is furanose form?
A furanose is a collective term for carbohydrates that have a chemical structure that includes a five-membered ring system consisting of four carbon atoms and one oxygen atom. The name derives from its similarity to the oxygen heterocycle furan, but the furanose ring does not have double bonds.
How many chiral carbons are present in the furanose form of fructose?
Fructose has got 16 optical isomers in total due to four chiral centers in it.
What is furanose and Pyranose?
Furanoses and Pyranoses Cyclic sugars that contain a five membered ring are called “furanoses”. The term is derived from the similarity with the aromatic compound furan and tetrahydrofuran. Cyclic sugars that contain a six membered ring are called “pyranoses”
How do you label carbons?
The numbers should be assigned so that the carbon with the functional group or branch has the LOWEST number. The numbers may go from either left to right or vica versa. Example top left – Pentenes: For alkenes or alkynes, only the first carbon of the double or triple bond need have a number.



