Which statement describes a chemical property of bromine
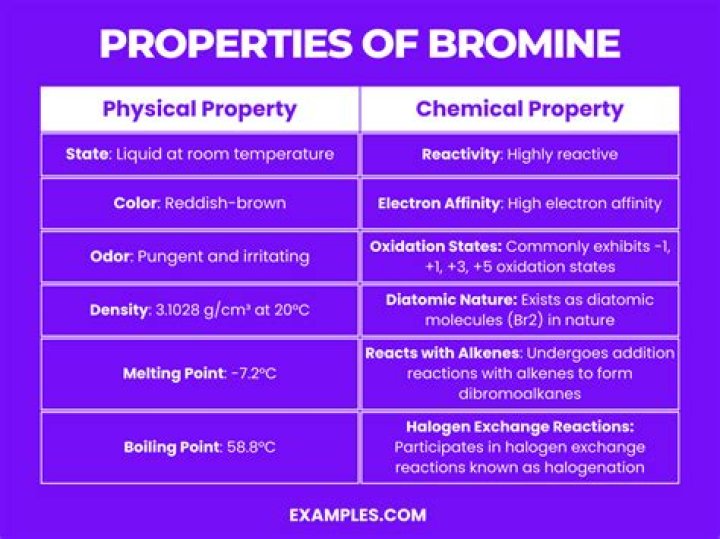
atomic number35melting point−7.2 °C (19 °F)boiling point59 °C (138 °F)specific gravity3.12 at 20 °C (68 °F)oxidation states−1, +1, +3, +5, +7
What are the chemical properties of bromine?
atomic number35melting point−7.2 °C (19 °F)boiling point59 °C (138 °F)specific gravity3.12 at 20 °C (68 °F)oxidation states−1, +1, +3, +5, +7
What is the chemical structure of bromine?
PubChem CID24408StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaBr2Synonyms7726-95-6 Br Bromine Dibromine molecular bromine More…
Which of the statement describes a chemical property?
A chemical property can be observed without changing the identity of the substance. A physical property is a characteristic that can be observed without changing the substance into another substance. The object reacts with acid to form water.What statement describes a chemical property of aluminum?
Aluminum is a soft and lightweight metal. It has a dull silvery appearance, because of a thin layer of oxidation that forms quickly when it is exposed to air. Aluminum is nontoxic (as the metal) nonmagnetic and non-sparking. Aluminum has only one naturally occurring isotope, aluminium-27, which is not radioactive.
What elements have similar physical and chemical properties bromine?
Fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) also exhibit similar properties to each other, but these properties are drastically different from those of any of the elements above.
Is bromine Reactive or nonreactive?
Chemical properties Bromine is a very reactive element. While it is less reactive than fluorine or chlorine, it is more reactive than iodine. It reacts with many metals, sometimes very vigorously.
Which statement describes a chemical property of sodium?
It’s a soft metal, reactive and with a low melting point, with a relative density of 0,97 at 20ºC (68ºF). From the commercial point of view, sodium is the most important of all the alkaline metals. Sodium reacts quickly with water, and also with snow and ice, to produce sodium hydroxide and hydrogen.What statement describes a chemical property of water?
Which statement describes a chemical property of water ? Under the proper conditions, water will breal apart into hydrogen and oxygen gas.
What is a chemical property quizlet?Chemical Property Definition. A characteristic that can only be observed during a chemical reaction and involves changing the substance into another substance. Physical Property Definition. A characteristic that can be observed or measured without changing the identity of the substance.
Article first time published onWhat type of element is bromine?
Bromine is a halogen, according to Chemicool. Halogen elements (fluorine, chlorine, bromine, iodine and astatine) are never found alone in nature and produce salts when reacting with metals, according to Chemistry Explained.
Which statement describes a chemical property of the element fluorine?
Fluorine is an univalent poisonous gaseous halogen, it is pale yellow-green and it is the most chemically reactive and electronegative of all the elements. Fluorine readily forms compounds with most other elements, even with the noble gases krypton, xenon and radon.
Which describes a chemical property of gasoline?
You would react with oxygen from the air and produce carbon dioxide and water vapor. All right, so, the chemical property of gasoline is the fact that it ignites easily.
Which statement describes a chemical property of iron iron oxidizes?
QuestionsAnswerExplanations7 Which statement describes a chemical property of iron? (1) Iron oxidizes. (2) Iron is a solid at STP. (3) Iron melts. (4) Iron is attracted to a magnet.1look for a reaction, not a phase change
Why is bromine so reactive?
For bromine or iodine to react, each atom needs to gain an electron to fill up its shell so that it is in a more stable state. Since bromine has fewer shells, its outer shell is closer to the nucleus so, there is a stronger force of attraction of the positively charged nucleus on the outer shell of electrons.
Which Dihalogen is most reactive?
Fluorine is the most reactive of the halogens and, in fact, of all elements, and it has certain other properties that set it apart from the other halogens. Chlorine is the best known of the halogen elements.
What elements does bromine bond with?
Bromine is a member of the halogen family of elements. Its companions include fluorine, chlorine, and iodine.
What elements have the same chemical properties?
Two different elements have similar chemical properties when they have the same number of valence electrons in their outermost energy level. Elements in the same column of the Periodic Table have similar chemical properties.
Which of the following elements would have similar chemical properties?
For example, pairs lithium (Li) and magnesium (Mg), beryllium (Be) and aluminium (Al), boron (B) and silicon (Si), etc are exhibiting similar properties. Boron and Silicon are both semiconductors. Thus Magnesium and Lithium have similar properties.
Which list consists of elements that have the most similar chemical properties?
The elements that have the most similar chemical properties are those in the same group, or column of the periodic table.
Which describes a chemical property of oxygen?
In normal conditions oxygen is a colourless, odourless and insipid gas; it condensates in a light blue liquid. Oxygen is part of a small group of gasses literally paramagnetic, and it’s the most paramagnetic of all. Liquid oxygen is also slightly paramagnetic.
Which property describes a chemical property of silicon?
Silicon is the most abundant electropositive element in The Earth’s crust. It’s a metalloid with a marked metallic luster and very brittle. It is usually tetravalent in its compounds, although sometimes its bivalent, and it’s purely electropositive in its chemical behaviour.
Which of the following describe a chemical property of gold?
Gold is also termed to be chemically “inert” hence, it reacts very sluggishly when reacting to other substances. For example gold exhibits no reaction to air and water and also does not itself corrode when exposed to long periods of harsh weathering conditions.
What is the chemical properties of salt?
Salt is white, odorless and it has a salty taste. All potassium (K), ammonium (NH4+) and sodium (Na) salts are soluble in water (H2O). Nitrites, nitrates, and bicarbonates can be dissolved in water. All metallic oxides, metallic carbonates, hydroxides, phosphates, and sulphides are insoluble in water.
What are two physical properties of sodium?
Atomic number11Melting point97.82°C (208.1°F)Boiling point881.4°C (1618°F)Volume increase on melting2.70%Latent heat of fusion27.0 cal/g
Which property is a chemical property of a substance quizlet?
the ability of a substance to burn is chemical property that involves a substance reacting quickly with oxygen to produce light and heat. the process is called burning. the ability of a substance to rust is a chemical property that involves a substance reacting slowly with oxygen. the process is called rusting.
How do chemical properties differ from physical properties quizlet?
What is the difference between chemical properties and physical properties? chemical properties are characteristics that describe a substance’s ability to change into different substances, and physical properties are characteristics that can be observed without changing it into another substance.
Why do all atoms of bromine have the same chemical properties?
Different isotopes of an element generally have the same physical and chemical properties because they have the same numbers of protons and electrons.
What is the element bromine used for?
Bromine is used in many areas such as agricultural chemicals, dyestuffs, insecticides, pharmaceuticals and chemical intermediates. Some uses are being phased out for environmental reasons, but new uses continue to be found. Bromine compounds can be used as flame retardants.
How is bromine used in agriculture?
Bromine compounds are effective pesticides, used both as soil fumigants in agriculture, particularly fruit-growing, and as a fumigant to prevent pests from attacking stored grain and other produce. … Bromine compounds are also used as intermediates to make some of the agriculture chemicals we take for granted.
What type of bond is formed in Br2?
A bond between two atoms of the same element is covalent. (H2, Cl2, Br2, I2, etc), and it is nonpolar. A bond between atoms of two different elements will be polar covalent or ionic.



