Which two substances bind using a lock and key mechanism
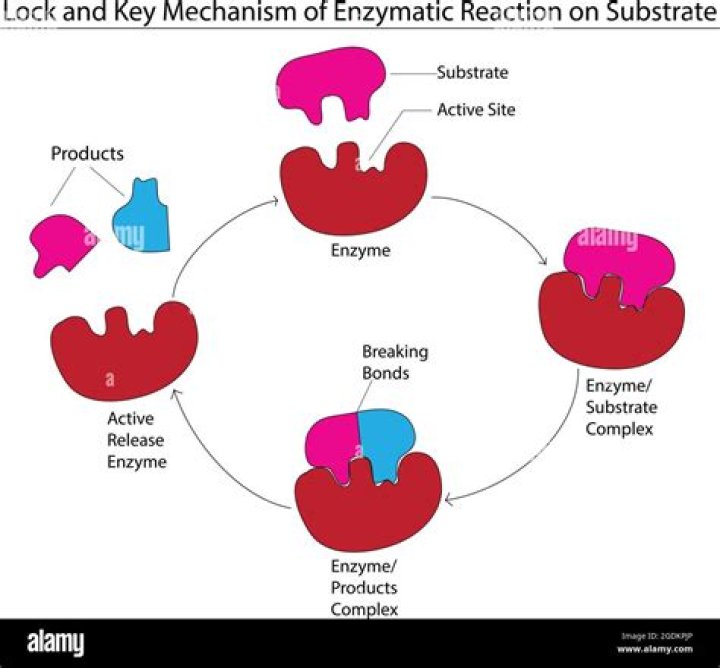
The lock and key mechanism is a metaphor to explain the specificity of the enzymes active site and the substrate. In the same way only certain keys fit a lock, only certain substrates fit an enzyme’s active site. The substrate is specific to a certain active site.
What does the lock and key mechanism refer to?
The lock and key mechanism is a metaphor to explain the specificity of the enzymes active site and the substrate. In the same way only certain keys fit a lock, only certain substrates fit an enzyme’s active site. The substrate is specific to a certain active site.
Which statement best describes how a catalyst can speed up chemical reaction?
Option a is the correct answer. A catalyst provides an alternative reaction pathway that has lower activation energy.
Which components is released from the active site of an enzyme during a chemical reaction?
The correct answer is product.How does a catalyst work?
A catalyst is a substance that increases the rate of a chemical reaction without being consumed in the reaction. A catalyst works by providing a different pathway for the reaction, one that has a lower activation energy than the uncatalyzed pathway.
Which component is released from the active site of?
Answer: The correct answer is Product. Enzymes are the bio-catalysts which help in increasing the rate of bio-chemical reaction taking place in a organism. Enzymes have specific regions for the binding of the substrate or reactant and is called as active site.
How is a lock and key like an enzyme and substrate?
Enzymes are highly specific. They must bind to a specific substrate before they can catalyze a chemical reaction. Like a key into a lock, only the correct size and shape of the substrate (the key) would fit into the active site (the key hole) of the enzyme (the lock). …
What is a substance that influences the reaction but does not participate in the reaction?
The activation energy must be reached. A substance that influences the reaction but does not participate in the reaction is a . catalyst. The lock-and-key mechanism refers to. the complementary shapes of an enzyme and a substrate.How do catalysts work quizlet?
How does a catalyst work? –To turn reactants into products activation energy is required. -If molecules do no posses enough energy when they collide they will not react. -A catalyst provides an alternative route for the reaction to take place with a lower activation energy.
How do catalysts work to accelerate chemical reactions quizlet?Catalyst speed up the reaction by lowering the activation energy or providing an alternative pathway for the reacting particles. … Concentration: Increasing concentration increases rate of reaction.
Article first time published onWhich reaction is an example of heterogeneous catalysis?
Which reaction is an example of heterogeneous catalysis? Ethene gas reacts with hydrogen gas by using a nickel catalyst.
What overall effect does adding a catalyst have on a chemical reaction?
What overall effect does adding a catalyst have on a chemical reaction? It lowers the activation energy that is needed.
Where are catalysts used?
Catalysts are used in industries to break down pulp to produce sanitary paper, to turn milk into yogurt, and to refine crude oil into a series of end products, among countless other uses. When a catalyst is used, a polluting chemical reaction can be reduced or replaced with an environment-friendly one.
Is a reactant that binds to a catalyst?
Reactions must take place very quickly, so they usually need a catalyst. Enzymes are catalysts for chemical reactions in living things. … Each enzyme binds a particular reactant, or substrate. The substrate fits into a part of the enzyme, like a key fits into a lock.
Is an enzyme a catalyst?
A fundamental task of proteins is to act as enzymes—catalysts that increase the rate of virtually all the chemical reactions within cells.
Why key and lock concept describes enzyme?
because the enzyme’s action parallels the action of a lock into which is fitted the key (substrate). If the key and lock do not match, the action does not work. It is the same with enzymes and substrates.
What is the product in the lock and key model?
Answers. The lock-and-key model portrays an enzyme as conformationally rigid and able to bond only to substrates that exactly fit the active site. The induced fit model portrays the enzyme structure as more flexible and is complementary to the substrate only after the substrate is bound.
Does the lock and key model explain inhibitors?
This modified lock and key model, known as the induced fit theory, also explains why some substrates, known as inhibitors, fit in the enzyme site but don’t create a chemical reaction.
What is the main difference in the lock and key and induced fit models of enzyme substrate binding quizlet?
Terms in this set (18) Who proposed the theory of the induced fit model? What is the difference between the lock and key model and induced fit? Lock and Key states that there is no change needed and that only a certain type will fit. However induced fit says the active site will change to help to substrate fit.
What is enzyme explain the difference between lock and key model and induced fit model of enzyme?
Explanation: The lock and key model states that the active site of an enzyme precisely fits a specific substrate. The induced fit model states that the active site of an enzyme will undergo a conformational change when binding a substrate, to improve the fit.
What is a substrate quizlet?
Define substrate. A substance that interacts with an enzyme. Define product. Anything that is produced as a result of a reaction.
What is the substrate of lactose?
without or with Enzyme A naturally occurring substrate of lactase is lactose (milk sugar) which is found in concentrations of up to 5 % in the milk of cows. Lactose is a disaccharide, made up of β- Page 3 4-3 galactose and glucose (Figure 4-3).
Which statement best illustrates a biotic or an abiotic factor that is often found in a city park quizlet?
Which statement best illustrates a biotic or an abiotic factor that is often found in a city park? Decaying leaves are an abiotic factor because they are not living.
What is a catalyst quizlet chemistry?
Catalyst. a substance that increases the rate of a chemical reaction without itself undergoing any permanent chemical change. Enzyme.
What occurs in a chemical reaction?
Chemical reactions occur when chemical bonds between atoms are formed or broken. The substances that go into a chemical reaction are called the reactants, and the substances produced at the end of the reaction are known as the products.
Which of the following best describes how a catalyst works?
Which of the following best describes how a catalyst works? It speeds up a reaction by providing a lower E a path, thereby increasing the number of reactant collisions with sufficient energy.
What effect does a catalyst have on the mechanism of a reaction?
A catalyst is a substance that can be added to a reaction to increase the reaction rate without getting consumed in the process. Catalysts typically speed up a reaction by reducing the activation energy or changing the reaction mechanism.
What two conditions of reactants must be met for a reaction to proceed?
- Molecules must collide.
- Molecules must collide with enough energy to begin to break the old bonds so new bonds can form. ( Remember activation energy)
- Molecules must collide with the correct orientation.
Is a substance which alters the rate of a reaction but itself does not undergo any permanent change at the end of the reaction?
catalyst A substance that increases the rate of a chemical reaction without itself undergoing any permanent chemical change.
What does the lock and key mechanism refers to?
The lock and key mechanism is a metaphor to explain the specificity of the enzymes active site and the substrate. In the same way only certain keys fit a lock, only certain substrates fit an enzyme’s active site. The substrate is specific to a certain active site.
When the enzyme and substrate binds it is called the?
The part of the enzyme where the substrate binds is called the active site (since that’s where the catalytic “action” happens). … Thanks to these amino acids, an enzyme’s active site is uniquely suited to bind to a particular target—the enzyme’s substrate or substrates—and help them undergo a chemical reaction.