Who is in the IRB
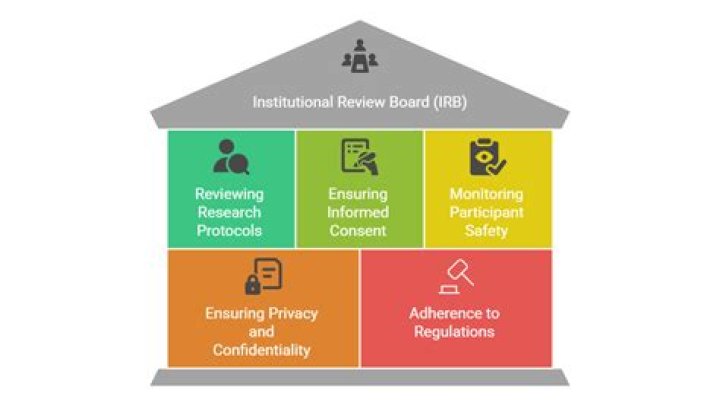
An Institutional Review Board (IRB) is a committee made up of individuals who have training in scientific areas, individuals who have expertise and training in non-scientific areas, and members of the community who may represent people who would participate as subjects in research studies.
How are IRB members selected?
The IRB Chair and IRB Director are responsible for selecting members to serve on the IRB. The selection process is conducted in consultation with the Institutional Official, Deans, Department Chairs, and other IRB members. The Institutional Official will appoint the IRB members.
Who serves on institutional review boards quizlet?
An IRB consists of a group of scholars from various departments who are mandated to review proposals to ensure that they comply with ethical guidelines and write a summary of proposed risks to subjects.
How many members are in the IRB?
Sec. 56.107 IRB membership. (a) Each IRB shall have at least five members, with varying backgrounds to promote complete and adequate review of research activities commonly conducted by the institution.What does an IRB committee do?
The IRB is charged with the responsibility of reviewing, prior to its initiation, all research (whether funded or not) involving human participants. The IRB is concerned with protecting the welfare, rights, and privacy of human subjects.
Which statement best describes the role of an IRB?
Which statement best describes the role of an IRB: a committee that reviews different types of human subjects research.
Do IRB members get paid?
Affiliated IRB Committee members do not receive any direct monetary compensation for participation on the board. Unaffiliated IRB Committee members will be reimbursed at an amount not to exceed $60 per month to pay for internet access and parking. Reimbursement payments will be issued quarterly.
What does the IRB do in psychology?
Institutional Review Boards (IRBs) are federally-mandated, locally-administered groups charged with evaluating risks and benefits of human participant research at their institution.What are the 4 important ethical issues IRB guidelines address?
- Respect for persons: respect for patient autonomy.
- Beneficence: maximize benefits and minimize harm.
- Justice: Equitable distribution of research burdens and benefits.
The IRB reviews research that involves human participants. Research involving animals must be reviewed by the Institutional Animal Care and Use Committee (IACUC) before it begins.
Article first time published onWhy might a researcher debrief his participants?
You submit a study for approval by the institutional review board (IRB), and they tell you that written informed consent is required. … Why might a researcher debrief his participants even if his study didn’t include any deceptive elements? To ensure that his participants had a good research experience. Dr.
What is the role of the institutional review board quizlet?
Institutional review boards (IRBs) are tasked with reviewing all studies involving human subjects to protect their rights and welfare.
What is the main goal of the Institutional Review Board IRB approval quizlet?
Institutional review board (IRB) definition: Committee that review, approve, and monitor clinical trial. Primary goal of IRB: Protect the right and safety of human subjects who participate in clinical trial.
What is an IRB protocol?
The protocol serves as the recipe for the conduct of the research activity. It needs to communicate all of the information that the following groups need to conduct their part of the study: the investigator and the investigative team, the data manager, the statistician and the IRB or other review body.
What is the purpose of doing a literature review?
A literature review establishes familiarity with and understanding of current research in a particular field before carrying out a new investigation. Conducting a literature review should enable you to find out what research has already been done and identify what is unknown within your topic.
What is IRB clinical trial?
Under FDA regulations, an Institutional Review Board is group that has been formally designated to review and monitor biomedical research involving human subjects. … Comprehensive list of regulations governing human subject protection and the conduct of clinical trials.
What is an IRB chair?
Institutional Review Board Advisory Committee (IAC) The IRB Chair, in addition to other IRB administration representatives from the Clinical Research Community, serves as a voting member on the IRB Advisory Committee (IAC) and will identify new information that might affect the HRPP, including laws, regulations, …
What types of research require IRB approval?
FDA regulations generally require IRB review and approval of research involving FDA-regulated products (e.g., investigational drugs, biological products, medical devices and dietary supplements) (21 CFR Part 56).
Is Institutional Review Board capitalized?
Many simply capitalize the term “Institutional Review Board” as the proper name of their instance. Regardless of the name chosen, the IRB is subject to the FDA’s IRB regulations when studies of FDA-regulated products are reviewed and approved.
What is the main focus of NIH's conflict of interest policy?
A conflict of interest policy is used to outline procedures for employees when a possible conflict exists between their own personal interests and the interests of the organization. The main focus of NIH’s conflict of interest policy is basically to prevent financial conflicts of interest.
Which justification would an IACUC most likely?
Which justification would an IACUC most likely accept if a research team plans to withhold pain relief (analgesia) from animals during a project? Documented evidence that the analgesic agent will interfere with the aims of the project.
What are the three principles discussed in the Belmont Report?
Three basic principles, among those generally accepted in our cultural tradition, are particularly relevant to the ethics of research involving human subjects: the principles of respect of persons, beneficence and justice.
What is justice in the Belmont Report?
Justice: This principle advocates fair treatment for all and a fair distribution of the risks and benefits of the research. It forbids exploitation of vulnerable people (for instance, economically disadvantaged or those with limited cognitive capacity) or those who are easily manipulated as a result of their situation.
How does IRB handle ethical cases?
The purpose of the IRB is to ensure that the investigator complies with the protocol and to demonstrate that the trial is necessary and that the risk-benefit ratio is acceptable by reviewing key trial documents to ensure that the subjects’ rights and well-being are protected.
What are the principles of research?
The four basic principles of research are classified as; autonomy, beneficence, non-maleficence, and justice.
What does the APA do?
Our mission is to promote the advancement, communication, and application of psychological science and knowledge to benefit society and improve lives. We do this by: Utilizing psychology to make a positive impact on critical societal issues.
What is the Iacuc in psychology?
acronym for Institutional Animal Care and Use Committee.
Which IRB is used for behavioral research?
The Behavioral/NonMedical IRB (IRB02) is responsible for reviewing and monitoring a subset of the research with human subjects conducted at the University of Florida.
What is the difference between an IRB and an Iacuc?
Research involving live vertebrate animals (except for humans) must be approved in advance by the Institutional Animal Care and Use Committee (IACUC). Research involving human participants may require approval of the Institutional Review Board (IRB).
How does a study become unethical in nature?
U.S. regulations that require an equitable selection of research subjects imply that a study that is otherwise ethical (e.g., a study with an acceptable risk-benefit ratio and whose subjects have freely consented) becomes unethical when it unfairly draws its research population from persons disadvantaged by reason of …
What is a human participant?
definition of a human participant is a living individual about. whom an investigator conducting research obtains (1) data or. samples through intervention or interaction with individual(s), or (2) identifiable private information.