Why do nonmetals gain electrons to form negative ions
The outer shells of non-metal atoms gain electrons when they form ions: the ions formed are negative, because they have more electrons than protons. the ions formed have full outer shells.
Why do non metals become negative ions?
Nonmetals form negative ions by receiving electrons from other atoms, giving them an overall negative charge and forming ions called anions.
Do metals or nonmetals tend to form negative ions?
Key Concepts and Summary Thus, nonmetals tend to form negative ions. Positively charged ions are called cations, and negatively charge ions are called anions. Ions can be either monatomic (containing only one atom) or polyatomic (containing more than one atom).
Why do nonmetals gain electrons?
Nonmetals want to gain electrons because they have more valence electrons than metals, so it is easier for them to gain electrons than lose the valance electrons to fulfill a stable octet. In addition, nonmetals’ valance electrons are closer to the nucleus, thus allowing more attraction between the two.Do nonmetals have positive or negative ions?
Cations (positively-charged ions) and anions (negatively-charged ions) are formed when a metal loses electrons, and a nonmetal gains those electrons. The electrostatic attraction between the positives and negatives brings the particles together and creates an ionic compound, such as sodium chloride.
Why do metals become positive and nonmetals become negative?
Ionic bonds form when metals and non-metals chemically react. By definition, a metal is relatively stable if it loses electrons to form a complete valence shell and becomes positively charged. Likewise, a non-metal becomes stable by gaining electrons to complete its valence shell and become negatively charged.
Why do nonmetals gain electrons when bonding with metals?
Nonmetals tend to gain electrons in order to achieve a full outer shell, so they are said to have high electronegativities. Alkaline metals, for example, would find it much easier to lose electrons than gain electrons, so they are not very electronegative.
Why do metals lose electrons and nonmetals gain electrons in the formation of ionic bonds?
Ionic bonds form only between metals and nonmetals. That’s because metals “want” to give up electrons, and nonmetals “want” to gain electrons. It takes energy to remove valence electrons from an atom and form a positive ion.Why do metals typically form positive ions and nonmetals typically form negative ions?
Ionic bonds form only between metals and nonmetals. That’s because metals “want” to give up electrons, and nonmetals “want” to gain electrons. It takes energy to remove valence electrons from an atom and form a positive ion. Energy is released when an atom gains valence electrons and forms a negative ion.
Why do metals and nonmetals usually form ionic compounds whereas two bonded nonmetals are never ionic?Why do metals and nonmetals usually form ionic compounds, whereas two bonded nonmetals are never ionic? … The difference in electronegativity between metals and nonmetals is high, meaning that it’s very easy for the very electronegative nonmetals to take electrons from non-electronegative metals.
Article first time published onWhy do metals always lose electrons to nonmetals?
Metal atoms lose electrons to nonmetal atoms because metals typically have relatively low ionization energies. Metals at the bottom of a group lose electrons more easily than those at the top. That is, ionization energies tend to decrease in going from the top to the bottom of a group.
Which ion is formed by metals by losing electrons to nonmetals?
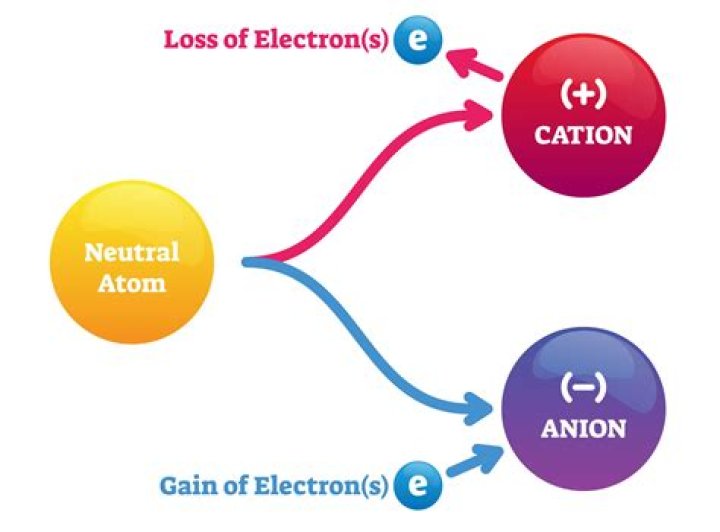
metal atoms lose electrons to form positive ions (cations ) non-metal atoms gain electrons to form negative ions (anions )
When atoms gain or lose electrons they become negatively or positively charged these negatively or positively charged atoms are known as?
An atom that loses one or more valence electrons to become a positively charged ion is known as a cation, while an atom that gains electrons and becomes negatively charged is known as an anion.
Why do two nonmetals form covalent bonds?
Nonmetals can form different types of bonds depending on their partner atoms. Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. … Atoms form covalent bonds in order to reach a more stable state.
Why do compounds of metals and nonmetals consist of ions?
The electronegativity differences between metals and most nonmetals are high. Therefore, the bonds have an ionic character. This makes sense with respect to ionization energy and electron affinity; the metal atoms are willing to lose electrons, and the nonmetal atoms are willing to gain them.
What type of charges do nonmetals develop in ionic compounds?
Nonmetals form negative ions (anions). A nitrogen atom must gain three electrons to have the same number of electrons as an atom of the following noble gas, neon. Thus, a nitrogen atom will form an anion with three more electrons than protons and a charge of 3−.
How do metals and nonmetals lose and gain electrons?
In a reaction between metals and nonmetals, metals generally lose electrons to complete their octet and non-metals gain electrons to complete their octet. Metal atoms lose electrons from their outer shell when they form ions: the ions are positive, because they have more protons than electrons.
What do nonmetals become after they accept electrons?
In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion.
How do metals and nonmetals differ in terms of how they lose gain electrons?
Metals tend to have 3 or less valence electrons. How do metals and nonmetals differ in terms of how they lose/gain electrons. Metals tend to lose electrons and nonmetals tend to gain electrons. … Halogens tend to gain one electron so that they can get a full outer level and be stable.
What do nonmetals do to form ions?
Nonmetals form negatively charged ions, or anions. They do this because they need to gain one to three electrons in order to achieve an octet of valence electrons, making them isoelectronic with the noble gas at the end of the period to which they belong.
Do nonmetals tend to lose electrons?
Non-metals tend to gain electrons to attain Noble Gas configurations. The have relatively high Electron affinities and high Ionization energies. Metals tend to lose electrons and non-metals tend to gain electrons, so in reactions involving these two groups, there is electron transfer from the metal to the non-metal.
What happens when metals react with nonmetals?
When metals react with non-metals, electrons are transferred from the metal atoms to the non-metal atoms, forming ions. The resulting compound is called an ionic compound. In all of these reactions, the metal atoms give electrons to the non-metal atoms.
Why atoms gain and lose electrons?
Explanation: Atoms and chemical species lose or gain electrons when they react in order to gain stability. Thus, typically, metals (with nearly empty outer shells) lose electrons to non-metals, thereby forming positive ions. The number of electrons depends on their position on the Periodic table (in simple terms).
What happens when atoms lose or gain electrons?
Most atoms do not have eight electrons in their valence electron shell. … Atoms that lose electrons acquire a positive charge as a result because they are left with fewer negatively charged electrons to balance the positive charges of the protons in the nucleus. Positively charged ions are called cations.
When an atom loses an electron it becomes a negative ion?
An atom that gains or loses an electron becomes an ion. If it gains a negative electron, it becomes a negative ion. If it loses an electron it becomes a positive ion (see page 10 for more on ions).
Why do only nonmetals form covalent bonds?
Non metals attain stability when they attain nobility . i. e. Fully filled outer shell. In order to achieve that, they undergo covalent bonding by sharing of electrons.
Why do nonmetals form covalent chloride?
Correct Answer: (b) they can share electrons with chlorine. Explanation: Non-metals form covalent chlorides by sharing of electrons with chlorine.
What type of bond is formed between nonmetals?
In general, covalent bonds form between nonmetals, ionic bonds form between metals and nonmetals, and metallic bonds form between metals.



