Why does boiling point increase from sodium to Aluminium
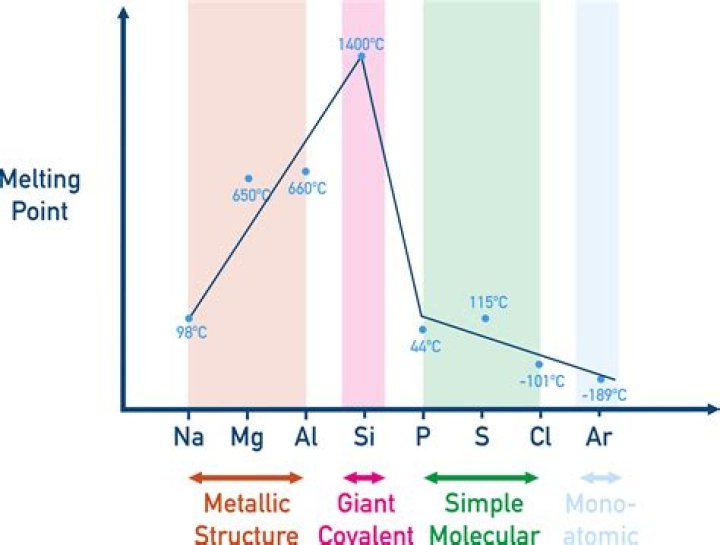
Melting and boiling points increase across the three metals because of the increasing strength of their metallic bonds. The number of electrons which each atom can contribute to the delocalized “sea of electrons” increases. The atoms also get smaller and have more protons as you go from sodium to magnesium to aluminum.
Why is there an increase in boiling point from sodium to aluminium?
This means there will be increased electrostatic attraction between electrons and ions as you go from Na to Al, therefore more energy is required to break the stronger metallic bonds, so boiling point increases.
Why does aluminum have higher melting point than sodium?
Thus, the atomic size of aluminium is smaller than that of sodium. -Decrease in atomic size leads to stronger bonds between the atoms of the metals. As a result more energy is required to break these bonds and cause them to melt. Thus, aluminium has a higher melting point than sodium.
Which statements help to explain the increase in melting point from sodium to aluminium?
We can now see that statement (C) — the number of delocalized electrons increases from sodium to aluminum — is true. And it partially explains why the melting point increases across this period. Statement (D) is also true; it is factually correct. The strength of the metallic bonding increases from sodium to aluminum.Why does aluminum have a high boiling point?
The positive Al cations are regularly arranged in a lattice and surrounded by a sea of free/delocalised negatively charged electrons (therefore metals can conduct electricity). The negative charge of electrons and positive charge of cations have strong attraction forces which require a lot of heat energy to break.
How does boiling point change across the periodic table?
Across the period from left to right of the periodic table size of atoms decreases. As the size decreases the strength of van der Waals forces also decreases. As strength decreases, the boiling point also decreases.
Why does sodium have a high boiling point?
It takes a lot of energy to overcome the strong electrostatic forces of attraction between oppositely charged ions, so ionic compounds have high melting and boiling points.
Why does the melting point increase as the atomic number increases?
1. As the atomic number of elements increases, the melting point increases because there are more electrons around the nucleus, which creates a stronger negatively-charged force. With stronger forces, the melting point rises.Why does sodium have a high melting point?
Sodium chloride has a high melting and boiling point There are strong electrostatic attractions between the positive and negative ions, and it takes a lot of heat energy to overcome them. Ionic substances all have high melting and boiling points.
Why does aluminium have a high melting point GCSE?Aluminium oxide (alumina, AlO) has a high melting point because it is a covalent crystal with strong AlO bonds. Here’s one of the 3 (alpha, beta, gamma) forms (Al grey, O red) : Aluminium [tri]bromide is molecular (AlBr) and the intermolecular forces are weak so it has a lower melting point.
Article first time published onWhy does the melting point of metals increase across Period 3 from sodium Na to aluminum AL?
Melting and boiling points increase across the three metals because of the increasing strength of their metallic bonds. The number of electrons which each atom can contribute to the delocalized “sea of electrons” increases. The atoms also get smaller and have more protons as you go from sodium to magnesium to aluminum.
Why does sodium have a lower melting point than magnesium?
According to electron sea theory magnesium shares two valence electrons in electron sea while sodium shares one suggesting that magnesium has stronger metallic bond than sodium so breaking metallic bond in magnesium will require more energy as compared to breaking metallic bond in sodium and that’s why melting point of …
Why does aluminum have a smaller atomic radius than sodium?
Of protons in aluminium(13) is more than sodium(11). That means the nuclear charge of aluminium is more and the electrons are pulled more ‘tightly’ as compared to sodium. Sodium on the other side has less nuclear charge as compared to aluminium and so the electrons are held comparatively loosely.
Why aluminium has higher melting point than magnesium?
Melting point of metals depends on the strength of the metallic bond. If aluminium has a higher melting point, it suggests that it has a stronger metallic bond than magnesium. Aluminium would have stronger attraction between its electrons and positive ions that keep the structure together and resistant against heat.
Why does Aluminium oxide have a higher melting point than sodium oxide?
For alumina, the ionic lattice is composed of Al3+ and O2− ions. Because the electrostatic force of attraction should be greater we would predict that alumina should have the greater melting point. The aluminum cation is also smaller than the sodium cation and thus more polarizing.
Does sodium has a high boiling point?
atomic number11atomic weight22.9898melting point97.81 °C (208 °F)boiling point882.9 °C (1,621 °F)specific gravity0.971 (20 °C)
Why does boiling point increase and then decrease across a period?
The melting and boiling points of the Group 1 elements decrease on descending the group. This is due to a decrease in the forces of attraction between the atoms. On crossing a period in the Periodic Table the atomic size decreases. On descending a group the atomic size increases.
Why does boiling point increase down the group?
Melting and Boiling Points (increases down the group) The melting and boiling points increase down the group because of the van der Waals forces. The size of the molecules increases down the group. This increase in size means an increase in the strength of the van der Waals forces.
Does boiling point increase as you go down the periodic table?
Different groups exhibit different trends in boiling and melting points. For Groups 1 and 2, the boiling and melting points decrease as you move down the group. For the transition metals, boiling and melting points mostly increase as you move down the group, but they decrease for the zinc family.
Why does boiling point increase with atomic number?
Lighter elements such as hydrogen and helium tend to have very low boiling points, and elements with greater atomic mass boil at higher temperatures. The atomic mass affects the forces between atoms, which in turn determine boiling points.
Why boiling point of noble gases increase down the group?
The boiling points of the noble gases increase as their atomic masses increase. The boiling points of the noble gases are very low compared to those of other substances with similar masses. … The dispersion forces increase as the size of the atom increases, so the boiling points also increase with atomic size.
Why does aluminium have a higher electrical conductivity than sodium?
Sodium, magnesium and aluminium They have metallic bonding, in which the nuclei of metal atoms are attracted to delocalised electrons. Going from sodium to aluminium: … there are more electrons that can move and carry charge through the structure … the electrical conductivity increases.
Why Aluminum has lower melting temperature than al2o3?
Hi, So my book says that MgO has the lower melting point than Al2O3. It explains this by saying that Aluminium is a very small ion with a large positive charge and so can approach the larger O2- and distort its electron cloud. This means that the bond has some added covalent character.
Why aluminium has higher boiling point than silicon?
The simplest answer to this question is that silicon has exactly one more proton than aluminum. As a result, silicon has a greater attraction (effective nuclear charge) for its valence electrons compared to aluminum. More energy is required to ionize an atom of Si when compared to Al.
Why is sodium metal so reactive?
Sodium (Na) is an alkali metal; it is shiny and soft with a single electron in its outermost shell. Alkali metals are highly reactive because they readily lose their outermost electron. Sodium combines with water in an explosive reaction.
Why is sodium named sodium?
A soft, silvery white and highly reactive metal, sodium was first isolated in 1807 by Humphry Davy during the process of electrolysis of sodium hydroxide. It’s symbol and name derive from the Latin Natrium or Arabicnatrun and the Egyptian word ntry (Natrun), all of which refer to soda or sodium carbonate.
What is the melting point and boiling point of aluminium?
AluminiumMelting point933.47 K (660.32 °C, 1220.58 °F)Boiling point2743 K (2470 °C, 4478 °F)Density (near r.t. )2.70 g/cm3when liquid (at m.p. )2.375 g/cm3
How can you increase the melting point of aluminum?
You can thus say that in order to increase the temperature of 1 g of aluminium by 1∘C , you need to supply it with 0.214 cal of heat. So, you now know that in order to increase the temperature of 5.5 g of aluminium by 1∘C , you need to supply it with 11.877 cal of heat.
Why melting and boiling point decreases down the group?
The melting/boiling point changes going down a group predominantly due to atomic radius. As a general rule for metals, a larger molecule means there is more shielding of nuclear charge and therefore bonding isn’t as strong, so the melting/boiling point is lower.
Why does melting point increase down Group 7?
In group 7, the further down the group an element is, the higher its melting point and boiling point . This is because, going down group 7: the molecules become larger. more energy is needed to overcome these forces.
Why does aluminium have a higher melting point than phosphorus?
The molecules are bigger than phosphorus molecules, and so the van der Waals attractions will be stronger, leading to a higher melting and boiling point.