Why does potassium permanganate spread in water
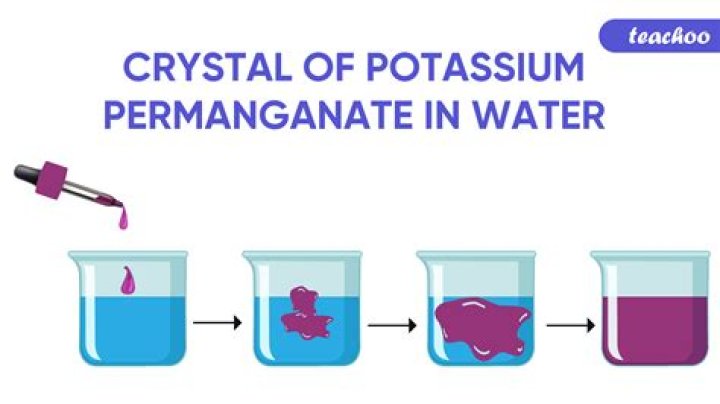
Because of the random movement of potassium permanganate particles, a dense purple solution forms in water at base of the beaker. The purple solution will slowly spread into the rest of the water throughout the beaker creating a less dense but evenly colored purple solution.
What happens when you put potassium permanganate crystals in water?
a) When potassium permanganate crystals are placed in hot water, the solution turns to purple in color because of a process called diffusion. Diffusion is the movement of molecules or atoms from high concentrated region to low concentrated region.
Why did the color move through the water?
The food coloring mixes through the hot water faster than it mixes with the cold water. This is because in hot water, the water molecules have more energy and are moving faster than the molecules of cold water.
Why does potassium permanganate spread faster in hot water?
Explanation: The kinetic energy of particles of water increases with increase in temperature. Therefore, the particles of hot water move faster. … Hence, potassium permanganate diffuses faster in hot water than in cold water.How is potassium permanganate used for water treatment?
Generally, a device injects potassium permanganate into the water in between the pump and holding tank. When treating a well, a concentration of 3.8 to 7.6 grams per gallon helps to remove iron bacteria. After the mixture has been fed into the well, agitation will make the treatment even more effective.
Why does potassium permanganate dissolve faster in hot water than in cold water?
The rate of diffusion is higher at higher temperatures.
How do you use potassium permanganate in water?
To achieve an appropriate dilution using potassium permanganate 0.1% solution, combine 1 part potassium permanganate with 10 parts hot water. Undiluted potassium permanganate has a striking purple color, but a diluted solution should be pink.
Why does diffusion increase with temperature?
The greater the difference in concentration, the quicker the rate of diffusion. The higher the temperature, the more kinetic energy the particles will have, so they will move and mix more quickly.What happens when potassium permanganate is dropped in hot water?
i) In a glass of cold water, the crystals calm down to the lowest while, just in the case of hot water, it slowly spreads. The crystals are often observed dissolving within the water just above and blue/ purple color is spread into the entire solution.
Why did the potassium permanganate crystals start to dissolve in water without being stirred?Why did the potassium permanganate crystals in Figure 7.12 on page 197 start to dissolve in water without being stirred or shaken? When potassium permanganate is placed in water its particles become more attracted to the water particles than to its own. This causes its particles to move apart.
Article first time published onWhat is the colour of water in which potassium permanganate is dissolved?
Answer: water with potassium permanganate dissolved in it is purple-pink in colour.
Does potassium purify water?
It successfully eliminates sulfur and manganese odors, which give well water its unpleasant smell. This compound also disinfects the water, preventing people from drinking harmful bacteria.
Is it safe to drink water with potassium permanganate?
Potassium permanganate can be used in drinking water in the prescribed quantity. It acts as a disinfectant and helps to remove harmful bacteria and germs. Potassium permanganate when added in excess may cause ill effects on health.
What is sodium permanganate used for in water treatment?
Sodium Permanganate is an oxidant used to control taste and odors, control biological growth in treatment plants, and remove iron and manganese. It can help to reduce the formation of disinfection byproducts (DBPs) by oxidizing precursors and reducing the demand for other disinfectants, such as chlorine.
What are the dangers of potassium permanganate?
* Breathing Potassium Permanganate can irritate the nose and throat. * Breathing Potassium Permanganate can irritate the lungs causing coughing and/or shortness of breath. Higher exposures can cause a build-up of fluid in the lungs (pulmonary edema), a medical emergency, with severe shortness of breath.
What happens if you eat potassium permanganate?
Side effects may include nausea, vomiting, and shortness of breath may occur. If a sufficiently large amount (about 10 grams) is eaten death may occur. Concentrated solutions when drunk have resulted in adult respiratory distress syndrome or swelling of the airway.
What process drives the movement of potassium permanganate particles?
This experiment demonstrates the principle of chemical diffusion using potassium permanganate in water. … Because of the random movement of potassium permanganate particles, a dense purple solution forms in water at base of the beaker.
Why do particles move down a concentration gradient?
A concentration gradient occurs when the concentration of particles is higher in one area than another. In passive transport, particles will diffuse down a concentration gradient, from areas of higher concentration to areas of lower concentration, until they are evenly spaced.
What causes rapid dispersion?
Extent of the concentration gradient: The greater the difference in concentration, the more rapid the diffusion. … Mass of the molecules diffusing: Heavier molecules move more slowly; therefore, they diffuse more slowly.
What factors affect the diffusion of water through a membrane by osmosis?
What factors affect the diffusion of water through a membrane by osmosis? The permeability of the membrane and the amount of water on each side of the membrane. If there is less water on one side, the water will diffuse to that side to even out the concentration of both sides.
Is used to help gasses dissolve into liquids?
liquid solvent, solute →gasmiscibilityusually very limited
Why is potassium Manganate purple?
The purple colour of the potassium permanganate is due to permanganate ion. Potassium permanganate acts a strong oxidizing agent and so it can be used to oxidize substances. The colour change changes occur in oxidation and reduction reaction mostly.
What is the nature of potassium permanganate and water?
Explanation: When the crystals of potassium permanganate are kept in water, the purple-coloured crystals of potassium permanganate break further into smaller particles that occupy the space between the molecules of water imparting a purple colour to the water. This is an example of diffusion.
Can you touch potassium permanganate?
Potassium permanganate crystals and concentrated solutions are caustic and can burn the skin. Even fairly dilute solutions can irritate skin and repeated use may cause burns.
Does potassium permanganate expire?
Expiry date: 12 months from the date of opening.
Why is my bath water pink?
Pink water is likely from potassium permanganate, a chemical used to oxidize iron and manganese. If too much is used your water will turn pink, and if far too much is used it will turn purple. As long as it is still pink it is not a danger to your health.
How do you remove potassium from water?
Potassium may be removed from water by means of reverse osmosis. Potassium is applied in water purification. For example, potassium permanganate is applicable for oxidation of waterborne compounds, such as for iron or manganese removal, and disinfection.
How do you neutralize potassium permanganate?
How to neutralize: Use 3% Hydrogen Peroxide to immediately neutralize (deactivate). Dose: 2 tbsp per 100 gallons (1 cup per 500 gal). Wait 4 days after neutralizing before treating again with Potassium Permanganate Solution.
What is the pH of sodium permanganate?
Standard Specifications: Sodium Permanganate (as NaMnO4), wt. % : 19.5 – 21.0. Specific Gravity, 25° C : 1.159 – 1.170. pH, Direct: 5.0 – 8.0.
How do you use potassium permanganate in a koi pond?
Dip: very effective for parasites, bacteria, ulcerations etc. Use 1 gram per 10 liter water for 7 minutes. Always aerate and ALWAYS remove the fish if it gets restless and jumpy. Control timing precisely and do not treat too many fish at the same time unless you can time the treatment of each fish correctly.



