Why does rock salt melt ice the fastest
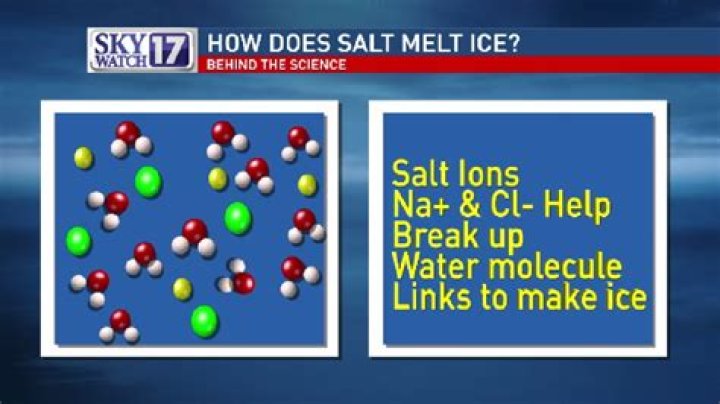
Because salt particles make it harder for water particles to freeze back onto the ice, the ice that is in contact with dissolved salt melts faster.
Why does rock salt melt ice faster than table salt?
Comparison: Table salt is more finely ground than rock salt. The finer something is, the more surface area it has. As a consequence, table salt will melt ice faster than rock salt simply because it has more surface area and so touches more of the ice at once.
Why does rock salt melt ice?
The actual reason that the application of salt causes ice to melt is that a solution of water and dissolved salt has a lower freezing point than pure water. … Ice in contact with salty water therefore melts, creating more liquid water, which dissolves more salt, thereby causing more ice to melt, and so on.
How fast does rock salt melt ice?
Conclusion. Universally, ice melt works faster than rock salt. Ice melt has a melting point of -25 degrees Fahrenheit, while rock salt melts at 25 degrees Fahrenheit and isn’t effective on surfaces below 10 degrees Fahrenheit. That being said, ice melt doesn’t provide instant traction on ice or snow.Which salt melts ice the fastest?
There are two types of salt traditionally used to melt ice on hard surfaces. One is rock salt, which has sodium chloride. Rock salt’s advantage is that it provides good traction. Salt with a combination of sodium chloride, calcium chloride and magnesium chloride melts ice faster, but it provides less traction.
Does table salt dissolve more rapidly than rock salt?
Solubility is affected by temperature, type of solute or solvent, particle size, and stirring. … They both dissolve in water, since salt particles are attracted to water particles, but table salt dissolves more quickly than rock salt.
How does rock salt melt ice?
After shoveling, apply the salt to frozen areas with a salt spreader or cup, following manufacturer’s instructions. If using your hands, make sure to wear gloves, as it can irritate bare skin. The ice will start to melt quickly, but thicker patches may take longer. Let it sit for a while to work.
What makes ice melt faster?
Salt will always melt ice quicker than both of them. … Salt, baking soda, and sugar will all act to lower the freezing point of the ice, making it melt quicker than the untouched ice cube.Can you eat rock salt?
Unless it’s labeled as edible, you can’t use it as an ingredient in food. Rock salt contains impurities, mostly minerals that are removed from salt that we use in our everyday cooking. … The salt forms a crust which will hold in moisture as the food cooks. It will also impart an evenly distributed salty taste.
How fast does salt melt ice?It starts to melt as quickly as straight calcium, but lasts as long as other sodium/potash blends. Below is a graph of melt volumes produced in 20 minutes at -10°C (14°F). An ice melter’s granule size and surface area both affect the melting process.
Article first time published onWhy does ice melt slower in salt water?
Assuming equal temperature, ice melts faster in salt water because salt water has a lower freezing point than fresh water, so the ice cube will have to absorb less heat in order to melt in salt water than in fresh water.
What salt melts ice the best?
Based on our research, calcium chloride is the best salt overall, though it’s a good idea to mix things up over the course of the season. Also, make a point of only applying the salt in the precise spots where you want to remove snow and ice.
Does Himalayan pink salt melt ice?
When ice needs to be melted by salt the fastest way possible pink himalayan salt will melt ice the fastest.
Why is sidewalk salt blue?
Blue colour has more visibility than other colours, so this colour is preferred, and there have been fewer complaints from residents thinking that their roads haven’t been salted. Not only do coloured salts reduce the complaints and visibility, but they also absorb more sunlight, making snow and ice removal easier.
Is rock salt good for snow?
Rock salt, also known as Halite, is the mineral form of sodium chloride. Salt lowers the freezing point of water. Small amounts of salt partially melt the ice forming a brine (solution of salt and water). … Rock salt works down to 5°F and helps provide instant traction on snow on ice.
Why does salt dissolve faster in hot water?
Energy from hot water molecules makes solids more soluble. In hot water, molecules are moving around more, so there are more collisions between the water molecules and a solid. … Most solids, including sugar and salt, become more soluble with increasing temperature.
Which dissolve faster rock salt or sugar explain?
What Happens. In this experiment, sugar should dissolve faster in solvents than salt does. The reason for this is because the sugar molecules are bigger than the ions of dissolved salt. This allows for more water molecules to surround a single particle, pulling it into solution faster.
Does salt dissolve in water without stirring?
A common misconception about dissolving is that heating and/or stirring are required for the dissolving process to occur. In this study, quantitative experimental evidence was collected and analyzed to demonstrate that neither heating nor stirring is required for dissolving.
What happens if you lick a salt rock?
There is no danger in licking the salt, after all, it is just salt,” Gaglione said. This was backed up by Patrik Ujszaszi of Himalayan Salt Factory, who wrote that licking a lamp “does not do any harm at all as the Himalayan salt has more natural minerals than the white table salt.”
Why is rock salt poisonous?
Calcium chloride, commonly referred to as rock salt, can be potentially harmful to pets, kids, and plants, and if you’re not careful, to adults as well. … This salts the soil and causes plants to absorb toxic levels of salt, leading to defoliation and/or plant death.
Why can't you eat ice cream salt?
Ice cream salt may contain potassium chloride, calcium chloride, and urea (carbonyl diamide). If consumed, it can irritate the gastrointestinal tract and abdominal pain.
Does salt make ice melt slower?
Adding salt to the ice/water mix causes a temperature drop that slows the melting rate and increases the freezing rate [3]. The net result is that the ice melts more and more slowly after the initial addition of salt. Why does salt melt ice? In pure water, at 0°C, ice melts just as fast as water freezes.
Which material melts ice the fastest?
Calcium Chloride Its ability to impose freezing point depression makes it an ideal material because it can accelerate the ice-melting process. The Peters Chemical Company says calcium chloride is its fastest ice-melting material.
What makes ice melt fastest conclusion?
Conclusions: Salt lowers the freezing/melting point of water/ice. When the salty ice cube was placed in the sun, the lower freezing point combined with continuous heat from the sun made the ice melt much faster.
Does rock salt stop ice from melting?
The saltwater ice will melt before the regular ice and as it does it will make the regular ice colder and thus stop it from melting. Salt actually makes ice melt at a lower temperature than the normal 32°F (0°C) and during the melting process it lowers the temperature of the ice.
Does salt actually melt ice?
In a nutshell, salt is a great ice melter because it causes “freezing point depression.” This means that salt helps in lowering the freezing point and, consequently, the melting point of water (the main component of snow and ice).
What melts ice the slowest?
Simple – the shape with the least surface area to volume is sphere. This, a spherical ice cube will melt the slowest.
Why does ice melt faster on metal?
Energy can be transferred (move) from the surroundings to the ice by conduction through the metal or plastic. Metal is a better conductor than plastic, so energy is transferred more quickly through the metal. This is why we saw the ice on the metal block melt more quickly.
Why does salt ice last longer?
As salt dissolves in water that chemical reaction requires energy in the form of heat and so it’ll draw that heat from the surrounding ice making it colder. … So adding salt to ice might make some of the ice melt faster, but it’ll lower the temperature of your cooler overall which can help to keep ice longer.
Does rock salt destroy concrete?
The answer is yes, salt does indirectly damage your concrete driveways, patios and sidewalks. Bumps and potholes don’t just appear due to regular wear and tear – salt damages concrete over time by causing corrosion to occur under the surface, leading to discolored, cracked and crumbling concrete.
Is rock salt and ice melt the same thing?
The difference lies in the mix. Halite, also known as rock salt, is strictly the mineral sodium chloride. Ice melt is typically a blend of sodium chloride with other minerals, such as magnesium and calcium.