Why is van der Waals radius greater than covalent radius
The main reason for why the van der Waals radius is greater than the covalent radius is that it does not take into account overlap. … When two atoms form a covalent bond, their electron clouds overlap. In essence, that is what a covalent bond really is – an overlap between orbitals.
What is the difference between van der Waals radius and covalent radius?
Covalent radius is half of the internuclear separation between the nuclei of two single-bonded atoms of the same species (homonuclear). While van der Waals radius is used to define half of the distance between the closest approach of two non-bonded atoms of a given element.
Why the metallic radius is greater than covalent radius?
Answer: Explanation: metal ions are touching the electrons and are not like overlapping which is the case of covalent radii. Because of this ,metallic radii of atoms is greater than the covalent radii of those metals..
Is van der Waals radius or ionic radius greater?
vdW radius (nm)ionic radius of X3- (nm)N0.1550.171P0.1800.212Why crystal radius is larger than covalent radius?
The measurement of the distance between the nuclei divided by two is then called the covalent radius for that atom. The metallic radii are measured in a different way. … Because of this the metallic radii of atoms will tend to be greater than the corresponding covalent radii of those metals.
What are Wonderwall radius and covalent radius of atom which one is smaller?
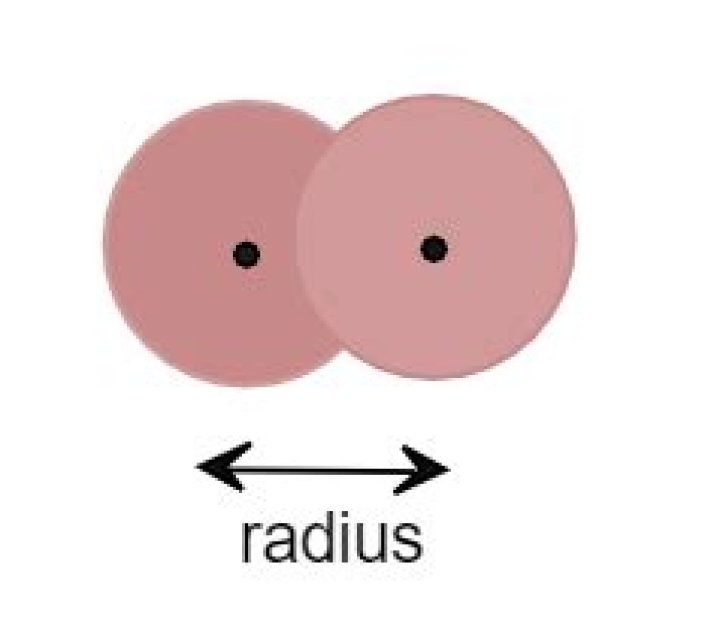
Clearly, one half of internuclear distance between adjacent atoms belonging to two neighbouring molecules will be more than one half of the distance between the nuclei of two like atoms forming a single covalent bond. Consequently, vander Waals‘ radii are always larger than covalent radii.
Why covalent radius is smaller than metallic radius?
The covalent radius is measured by overlapping of atomic clouds. Due to this the nuclei are pulled together as the electrons are positioned between the nuclei. This contraction of nucleus makes the covalent radius smaller. Metallic radii are measured as the distance between two metal atoms bonded with a single bond.
Is Van der Waals radius the same as atomic radius?
Definitions. Widely used definitions of atomic radius include: Van der Waals radius: In the simplest definition, half the minimum distance between the nuclei of two atoms of the element that are not otherwise bound by covalent or metallic interactions.Why Van der Waals radius is stronger than metallic and covalent radius?
Values of Van der Waals’ radii are larger than those of covalent radii because the van der Waals’ forces are much weaker than the forces operating between atoms in a covalently bonded molecule.
Why does the ionic radius increase down the group?As you move down a column or group, the ionic radius increases. This is because each row adds a new electron shell. … More protons are added, but the outer valence shell remains the same, so the positively charged nucleus draws in the electrons more tightly.
Article first time published onWhat is the difference between covalent radius and metallic radius?
The key difference between covalent radius and metallic radius is that the covalent radius is half the distance between two homonuclear atoms that are in a covalent bond, whereas the metallic radius is half the distance between two adjacent metal ions in a metallic structure.
What is Wonderwall radius?
The Van der Waals radius is equal to one half the distance between two unbonded atoms when the electrostatic forces between them are balanced. In other words, it is half of the closest distance between two atoms that aren’t bonded or within the same molecule.
Which one is greater covalent radius or ionic radius?
Correct among the following van der Waal’s radius is larger than covalent radius Covalent radius is larger than ionic radius Ionic radius is larger than metallic radius All of these.
Is covalent radii greater than atomic radii?
Covalent Radius Covalent radii will increase in the same pattern as atomic radii. The reason for this trend is that the bigger the radii, the further the distance between the two nuclei.
Which is greater ionic radius or metallic radius?
Metals – the atomic radius of a metal is generally larger than the ionic radius of the same element.
Why size of anion is larger than cation?
A cation has a fewer number of electrons than its parent atom, while its nuclear charge remains the same. As a result, the attraction of electrons to the nucleus is more in a cation than in its parent atom. … Hence, an anion is larger in radius than its parent atom.
Why does covalent radius increase down a group?
As you move down a group in the periodic table, the covalent radius increases. Atoms increase in size. This is because of the screening effect of the filled inner electron levels. These electrons screen or shield the outer electrons from the nuclear charge.
What is van der Waals radius Class 11?
– Van der Waals radius: It is half of the distance between two similar atoms in a separate molecule in a solid. -Ionic radius: A neutral atom charges to a cation by the loss of one or more electrons and an anion by the gain of one or more electrons.
Is metallic radius bigger than Van der Waals radius?
The Metallic radii are smaller than the Van der Waals‘ radii, since the bonding forces in the metallic crystal lattice are much stronger than the Van der Waal’s forces.
Why do noble gases not have measured covalent radius?
covalent radius is defined for those elements which forms a covalent bond. noble gases (like argon) don’t form any bond. so for them vanderwall radius is considered.
Why do atomic radii of noble gases larger in their periods?
The atomic radii of the noble gases are by far the largest in their respective periods. This is due to the fact in case of noble gas, since they exists as monoatomic gases. The radii is van waals radii, while in other it is covalent radii van der walls radii are larger than covalent radii.
Is van der Waals radius the largest?
Ne, O and F are the second period elements while Cl is 3rd period element so it has the largest van der waals radius.
Why do van der Waals forces increase with size of molecule?
– When a dipole molecule and polar molecules come closer to each other than the positive pole of the dipole attracts negative charge of polar molecules. … The strength of van der waal’s forces increases due to the increase in the number of electrons present and the size of the molecules.
What determines van der Waals radius?
Van der Waals radii may be determined from the mechanical properties of gases (the original method), from the critical point, from measurements of atomic spacing between pairs of unbonded atoms in crystals or from measurements of electrical or optical properties (the polarizability and the molar refractivity).
When comparing sizes of ions which ion has the largest radius and which ion has the smallest radius in an isoelectronic series?
K+, Cl−, and S2− form an isoelectronic series with the [Ar] closed-shell electron configuration; that is, all three ions contain 18 electrons but have different nuclear charges. Because K+ has the greatest nuclear charge (Z = 19), its radius is smallest, and S2− with Z = 16 has the largest radius.
Why is ionic radius smaller than atomic?
The ionic radius is half the distance between two gas atoms that are just touching each other. Values range from 30 pm to over 200 pm. … If the atom loses its outermost electron (positively charged or cation), the ionic radius is smaller than the atomic radius because the atom loses an electron energy shell.
Why is the radius of an atom larger than the radius of a positive ion formed from it?
When an atoms attracts extra electrons it becomes a negative ion. The negative ion is larger than the original atom. The positive nucleus remains the same, with the same attractive force. … This means with more electrons but the same number of positive protons, the size of the ionic radius will increase.
What is covalent radius and ionic radius?
Measurement of Radius Nonpolar atomic radii: The radius of an atom is derived from the bond lengths within nonpolar molecules; one-half the distance between the nuclei of two atoms within a covalent bond. … Crystal Radii: The atomic or ionic radius is determined using electron density maps fro X-ray data.
What is the van der Waals radius of hydrogen atom in water molecule?
The contact distance distributions and the two sets of radii are derived in a systematic and self-consistent manner using an iterative procedure. The Coulombic radii for polar atoms are, on average, 0.18 A smaller than their VDW radii. The VDW radius of water is 1.7 A, which is 0.3 A larger than its Coulombic radius.
Why are cations larger and anions smaller than their respective atoms?
In general, anions are larger than the corresponding neutral atom, since adding electrons increases the number of electron-electron repulsion interactions that take place. Cations are smaller than the corresponding neutral atoms, since the valence electrons, which are furthest away from the nucleus, are lost.