Do all polar molecules contain polar covalent bonds
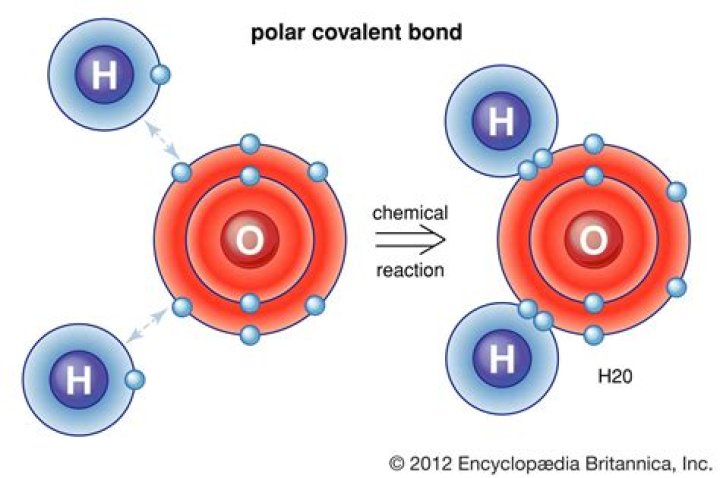
A polar covalent compound is one in which there is a slight difference in electric charge between opposite sides of the molecule. All polar compounds contain polar bonds. But having polar bonds does not necessarily result in a polar compound. It depends on how the atoms are arranged.
Can you have a non polar molecule with polar covalent bonds?
Often, the polarity of the bonds is the same as the polarity of the molecule. However, there are nonpolar molecules with polar bonds and polar molecules with nonpolar bonds! For example, boron trifluoride is a nonpolar molecule that contains polar covalent bonds.
Which molecule contains both polar and non polar covalent bonds?
Also, hydrogens are evenly distributed around carbon, so all the poles cancel out each other. Thus, $C{H_4}$is a nonpolar molecule. Hence, only ${H_2}{O_2}$ molecules have both polar and nonpolar bonds and hence the correct option is C.
Do polar molecules form covalent bonds?
Polar covalent molecules exist whenever there is an asymmetry, or uneven distribution of electrons in a molecule. One or more of these asymmetric atoms pulls electrons more strongly than the other atoms.How do you know if a bond is polar covalent?
The terms “polar” and “nonpolar” usually refer to covalent bonds. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; if the result is between 0.4 and 1.7, then, generally, the bond is polar covalent.
Are all covalent bonds molecules?
We know that a group of atoms joined by only covalent bonds is known as a molecule. It should be noted, however, that the word molecule should only be used in reference to covalent compounds.
Which molecules have polar covalent bonds?
- Water – H2O.
- Ammonia – NH. …
- Sulfur dioxide – SO. …
- Hydrogen sulfide – H2S.
- Ethanol – C2H6O.
Which does not contain polar bonds?
Hydrogen and carbon have similar electronegativity values, so the C—H bond is not normally considered a polar covalent bond. Thus ethane, ethylene, and acetylene have nonpolar covalent bonds, and the compounds are nonpolar.Which of the following is a polar molecule that has polar covalent bonds?
Hydrogen fluoride (HF) is another example of a molecule that has a polar covalent bond. Fluorine is the more electronegative atom, so the electrons in the bond are more closely associated with the fluorine atom than with the hydrogen atom.
Which type of bond is not present in hno2 molecule?In the above molecules , polar covalent bond is present between O and H and non polar covalent bond is present between N and O. Hence it lacks ionic and coordinate covalent bond.
Article first time published onWhich among the following are non polar molecules?
CH3Cl.
What is the electronegativity difference of a polar covalent bond?
A bond in which the electronegativity difference between the atoms is between 0.4 and 1.7 is called a polar covalent bond. A polar covalent bond is a covalent bond in which the atoms have an unequal attraction for electrons and so the sharing is unequal.
How can you tell the difference between polar and nonpolar covalent bonds?
If the electronegativity of two atoms is basically the same, a nonpolar covalent bond will form, and if the electronegativity is slightly different, a polar covalent bond will form.
Is PH3 polar covalent?
The electronegative property of PH3 found in the periodic table attracts shared pairs of valence electrons, creating covalent bonds. However, due to the unbonded electron, there will be asymmetrical charge distribution. With this, the PH3 is a polar molecule with nonpolar covalent bonds, not polar bonds.
Which of the following does not contain covalent bonds?
Sulphuric acid is the one which has both covalent bond as well as ionic bond. the oxygen is covalently bonded to the sulphur and the hydrogen is always bonded to the two oxygen atoms by the ionic bond. Hence \[KCl\] is one which does not have any covalent bond.
Which one of the following molecules has a non-polar covalent bond?
The correct answer is b. Carbon Dioxide (CO2) is a non-polar covalent compound because it has linear, symmetrical structure, with 2 oxygen atoms…
Where are covalent bonds found?
Covalent bonds are commonly found in carbon-based organic molecules, such as our DNA and proteins. Covalent bonds are also found in inorganic molecules like H2O, CO2, and O2. One, two, or three pairs of electrons may be shared, making single, double, and triple bonds, respectively.
Which of the following molecules contains polar covalent bonds only?
The molecule that contains a polar covalent bond is c. H-F because it consists of fluorine and hydrogen atoms which have electronegativity differences…
Does HNO3 have covalent bond?
The first bond is between the oxygen and Nitrogen, and it is called covalent bond since it is formed by two non-metals.
Does HNO3 have coordinate covalent bond?
Yes, Nitric acid (HNo3) is a coordinate bond.
Which type of bond is present in HNO3 molecule?
Nitric acid is a nitrogen oxoacid of formula HNO3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen atoms. It has a role as a protic solvent and a reagent. It is a conjugate acid of a nitrate.
Which of the following bonds is least polar?
The least polar bond would be between atoms have have the smallest difference in electronegativity. c. Br-Br: As they are two of the same atom, they will have no electronegativity difference, making the bond nonpolar.
In which of the following molecule is the covalent bond most polar?
HCl is most polar due to high electronegativity of Cl.
Which of the following is a polar molecule?
Water is a polar molecule due to the strong electronegativity of the oxygen atom. This forces most of the electrons to the side of the molecule where oxygen is present, creating a highly negative area.
How does electronegativity relate to polar and nonpolar covalent bonds?
Electrons in a polar covalent bond are shifted toward the more electronegative atom; thus, the more electronegative atom is the one with the partial negative charge. The greater the difference in electronegativity, the more polarized the electron distribution and the larger the partial charges of the atoms.
What must be always true if a covalent bond is polar?
A molecule is polar if the shared electrons are equally shared. A molecule is nonpolar if the shared electrons are are equally shared.
Is AsH3 polar covalent?
However, the molecular geometry of AsH3 looks like a tetrahedral and one lone pair on the top of the geometry. It’s the AsH3 molecule’s asymmetrical geometry. As a result, the AsH3 molecule is polar.
How many polar bonds does PH3 have?
(b) PH3: Phosphorus (Group 5A) has five valence electrons, and will complete its octet by making three bonds. There are three hydrogens, so it makes a single bond with each. This compounds is the phosphorus-containing analogue to ammonia.
Is PH3 covalent or ionic?
PH3 is a covalent polar compound. Phosphorous is bonded to three hydrogen atoms and has a lone pair of electrons. Since the electronegativity of phosphorous and hydrogen is nearly same so the covalent bond is non-polar.



