How do solutes affect boiling point
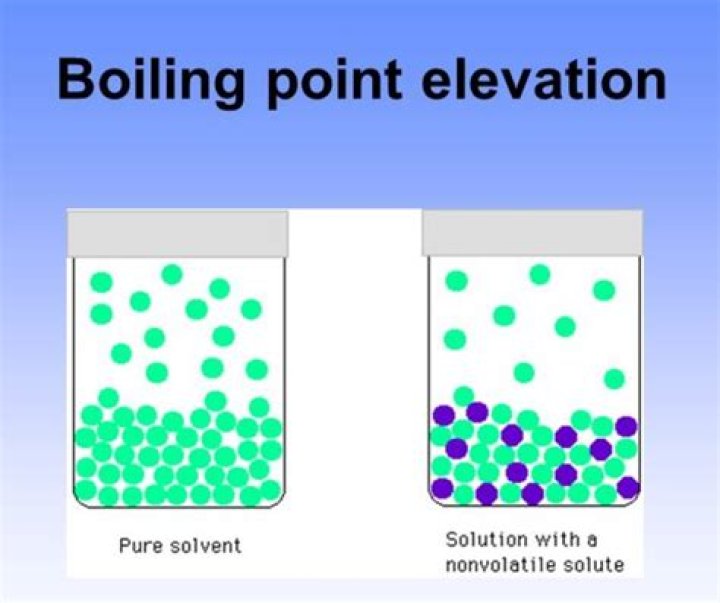
Because the presence of solute particles decreases the vapor pressure of the liquid solvent, a higher temperature is needed to reach the boiling point. This phenomenon is called boiling point elevation. For every mole of particles dissolved in a liter of water, the boiling point of water increases by about 0.5°C.
Why does boiling point increase when solute is added?
Even without a charged solute, adding particles to water raises the boiling point because part of the pressure the solution exerts on the atmosphere now comes from solute particles, not just solvent (water) molecules. The water molecules need more energy to produce enough pressure to escape the boundary of the liquid.
Does the addition of solutes to water increase or decrease its freezing point?
The presence of a solute lowers the freezing point of any solvent; this effect is called freezing-point depression. The key to understanding this effect is that the solute is present in the liquid solution, but not in the pure solid solvent. Example: think of pure ice cubes floating in salt water.
Do solutes decrease the boiling point of a solvent?
The effect of adding a solute to a solvent has the opposite effect on the freezing point of a solution as it does on the boiling point. A solution will have a lower freezing point than a pure solvent.How do solutes affect the boiling and freezing point of a solvent?
Boiling point elevation is the raising of a solvent’s boiling point due to the addition of a solute. Similarly, freezing point depression is the lowering of a solvent’s freezing point due to the addition of a solute. In fact, as the boiling point of a solvent increases, its freezing point decreases.
What effect does adding a solute have on the boiling point of a solution answers com?
The amount that the boiling point increases in the presence of solute can be calculated by using the boiling point elevation constant and the molality of the solution. The addition of solute dilutes the solvent molecules and makes it harder for them to escape into the gaseous phase.
How do solutes affect the properties of solvents?
Solutes can affect the taste, smell and appearance of the solvent. They can also affect physical properties like boiling or melting point, electrical resistance, and so on.
How does solute decrease freezing point?
Nonvolatile solutes lower the freezing point by blocking the solvent particles from congregating. … And thus, nonvolatile solutes make it harder to freeze, lowering the freezing point. The same solutes will also raise the boiling point.Why does increasing the solute concentration decrease the melting point?
The decrease in the vapor pressure of the solvent that occurs when a solute is added to the solvent causes an increase in the boiling point and decrease in the melting point of the solution. According to this figure, the solution can’t boil at the same temperature as the pure solvent.
Why does CS2 have a higher boiling point than CO2?CS2 has a higher boiling point than CO2 despite having similar intermolecular forces because it has a larger molar mass. … Intermolecular forces are generally stronger than bonding forces. Increasing the pressure on a solid usually causes it to become a liquid.
Article first time published onWhy is the boiling point of HBr less then the boiling point of Br2?
(c) Very polar – Hydrogen bonds between the molecules. … HCl is more polar, but the increased mass of HBr gives it a higher boiling point. Br2 (58.78 °C) and I2 (184.35 °C) Answer. Both are nonpolar, but I2 has more mass and it is more polarizable (stronger intermolecular forces), so it will have the higher boiling point …
Which has a higher boiling point HBr or HF?
We can conclude, the order of boiling point followed is HF > HI > HBr > HCl. … The reason is the presence of strong hydrogen bonding in the HF, as hydrogen intermolecular interactions are greater than the van der Waals forces. So, HF has a higher boiling point than the HI.
What effects do dissolved solutes have on the melting and boiling points of a substance?
When a solute dissolves in a liquid, the concentration of solvent decreases by dilution. Its mole fraction decreases, and both boiling point and freezing point depend on mole fraction. (They are related to the vapor pressure of the solid and liquid, also dependent on mole fraction.)
How does Van t Hoff factor affect boiling point?
Overall, boiling point elevation will be proportional to the moles of solute multiplied by the van’t Hoff factor. Using this proportion, we can find the solute that will most impact the boiling point of water.
How can solutes affect the physical properties of a solution such as freezing point and boiling point?
The effect of adding a solute to a solvent has the opposite effect on the freezing point of a solution as it does on the boiling point. … At a given temperature, if a substance is added to a solvent (such as water), the solute-solvent interactions prevent the solvent from going into the solid phase.
When some solutes are added to some solvent the viscosity?
When some solutes are added to some solvents, the viscosity of the resultant solution is found to be greater than that of the original solvent. However, ‘raising of viscosity’ is not a colligative property.
Why boiling point of solution is higher than solvent?
The boiling point of a solution, then, will be greater than the boiling point of the pure solvent because the solution (which has a lower vapor pressure) will need to be heated to a higher temperature in order for the vapor pressure to become equal to the external pressure (i.e., the boiling point).
Why is sio2 boiling point so high?
Melting and boiling points: The large structures (the metal oxides and silicon dioxide) have high melting and boiling points because a large amount of energy is needed to break the strong bonds (ionic or covalent) operating in three dimensions.
Why does BrCl have a lower boiling point than Br2?
The molecule of Br₂ is non-polar, and thus Br₂ molecules are held together by weak London dispersion forces. The molecule of BrCl is polar, and thus BrCl molecules are held together by stronger dipole-dipole attraction. Consider only this factor, Br₂ has weaker intermolecular forces than BrCl.
What effect does adding a solute have on boiling point of a solution apex?
This is true for any solute added to a solvent; the boiling point will be higher than the boiling point of the pure solvent (without the solute). In other words, when anything is dissolved in water, the solution will boil at a higher temperature than pure water would.
How does air pressure affect the boiling point of a liquid?
The pressure of gas above a liquid affects the boiling point. In an open system this is called atmospheric pressure. The greater the pressure, the more energy required for liquids to boil, and the higher the boiling point.
Do solutes lower melting point?
First, this only happens for dilute solutions. And in this case, it does not matter what you put in the solvent, the melting/freezing point will always decrease. The solute particles displace some solvent molecules in the liquid phase and therefore reduce the concentration of solvent.
What happens when the concentration of a solute increases?
The volume on the side with the solute increases until the number of water molecules on both sides is equal. Increasing the concentration of solute reduces the space available for water molecules, which reduces their numbers. This in turn increases the tendency of the water to flow into that side from the other side.
How does concentration of the solute affect dissolution?
Dissolution is the process where a solute in gaseous, liquid, or solid phase dissolves in a solvent to form a solution. Solubility is the maximum concentration of a solute that can dissolve in a solvent at a given temperature. At the maximum concentration of solute, the solution is said to be saturated.
Does adding solute increase vapor pressure?
When a solute is added to a solvent, the vapor pressure of the solvent (above the resulting solution) is lower than the vapor pressure above the pure solvent.
Why does the presence of a solute in a solvent depress its freezing point?
Solutions freezing points are lower than that of the pure solvent or solute because freezing, or becoming solid, creates order and decreases entropy. Solutions have high entropy because of the mix of solvent and solute, so it takes more energy to decrease their entropy to the same point.
Why does the presence of a solute affect the freezing point of the pure solvent?
Figure below shows the phase diagram for a pure solvent and how it changes when a solute is added to it. The solute lowers the vapor pressure of the solvent resulting in a lowering of the freezing point of the solution compared to the solvent.
Why does carbon disulfide have a low boiling point?
Carbon disulfide is made by the reaction of carbon and sulfur. … Carbon disulfide is denser than water and only slightly soluble in it. Its boiling point is 46.3° C (115.3° F) and its freezing point -110.8° C (-169.2° F); its vapour, which is heavier than air, is ignited with extraordinary ease.
Why does carbon disulfide have a high boiling point?
The London dispersion forces among molecules of CS2(l) are stronger because CS2 has a larger, more polarizable electron cloud than COS. These stronger intermolecular forces increase the boiling point of the substance (LO 2.11; SP 6.2, 6.4).
Which has a higher boiling point CO2 or CO?
The larger the small covalent molecule, the greater the intermolecular bonds, hence higher boiling / melting point. CO2 has 3 atoms involved in the molecule and is therefore larger than O2 that has 2 atoms. Hence, CO2 has a higher boiling / melting point compared to O2.
Why is Br2 have higher boiling point than BrCl?
However, due to the greater polarizability of the electron cloud of Br2 compared to that of BrCl, the London forces in Br2 (l) are stronger than the combined intermolecular forces in BrCl (l). Thus, the boiling point of Br2 (l) is greater than that of BrCl (l). 2.



