Is aspirin an ester
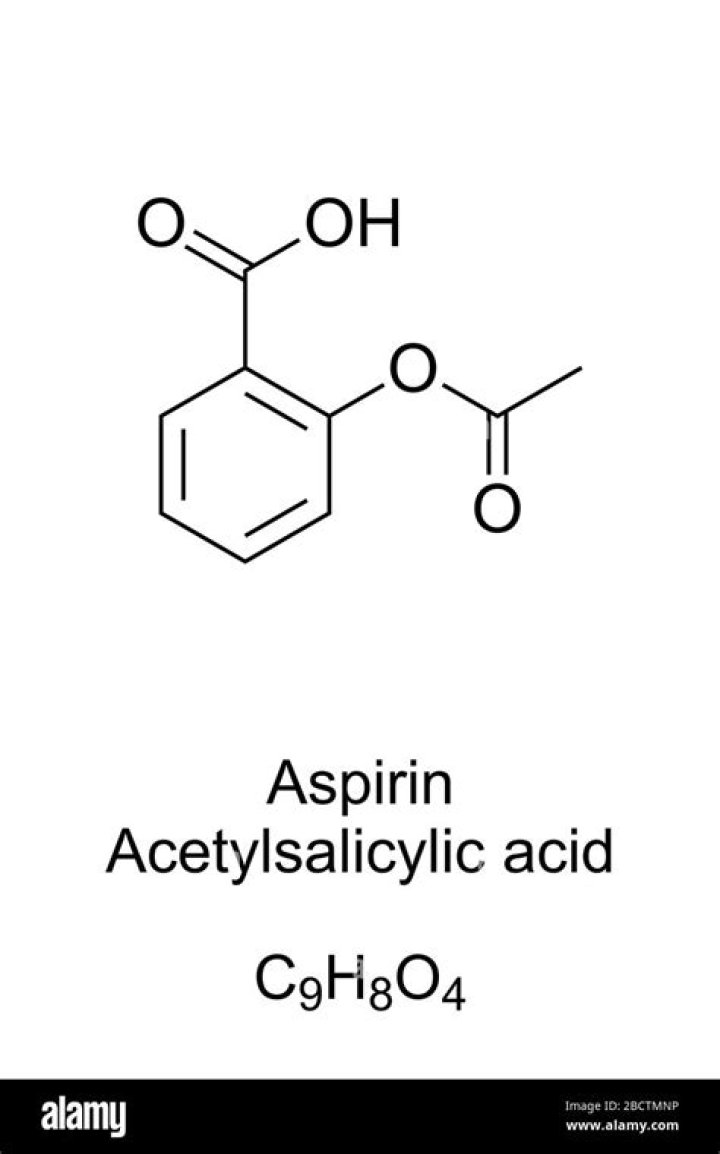
Aspirin is a trade name for acetylsalicylic acid, a common analgesic. Acetylsalicylic acid is an acetic acid ester derivative of salicylic acid.
Is aspirin made through esterification?
The preparation consists of an esterification reaction catalyzed by acid (H2SO4 or H3PO4), where salicylic acid treated with acetic anhydride gives acetylsalicylic acid (aspirin). In this reaction, a hydroxyl group is converted to an ester, with acetic acid as a byproduct.
Is salicylic acid an ester?
salicylic acid or 2-hydroxybenzoic acid, C6H4(OH)CO2H, a colorless, crystalline organic carboxylic acid that melts at 159°C; it is soluble in ethanol and ether but is only slightly soluble in water. … This ester hydrolyzes, not in the acidic stomach, but in the alkaline intestines, releasing free salicylic acid.
What kind of compound is aspirin?
Showing Compound Acetylsalicylic acid (FDB000894) Aspirin, also known as acetylsalicylate or easprin, belongs to the class of organic compounds known as acylsalicylic acids. These are o-acylated derivatives of salicylic acid. Aspirin has been detected, but not quantified in, herbs and spices.Why Is aspirin a carboxylic acid?
The common name of aspirin is acetylsalicylic acid and its IUPAC name is 2-Acetoxybenzoic acid. Between ester and carboxylic acid functional groups the highest priority functional group is carboxylic acid. Hence, in aspirin carboxylic acid is the principal functional group. Therefore, the correct option is B, -COOH.
Does aspirin have an ether?
In the case of aspirin, the ether group is a combination of a simple phenol group (OH) and acetic acid (C2H4O2). When the ether group in aspirin is hydrolyzed, acetic acid is formed, and the aspirin molecule becomes salicylic acid.
Is aspirin a phenol or ester?
Aspirin is an ester which passes through the stomach unchanged before being hydrolysed by the basic medium of the intestine to form the active compound. When a carboxylic acid reacts with an alcohol (such as ethanol or a phenol), the products are water and an ester.
Is aspirin a hydrocarbon?
Aspirin. Aspirin has proven itself one of the safest and most reliable medications over the past decades. … Yet most aspirin manufacturing begins with benzene, a hydrocarbon that is typically derived from petroleum products.Is aspirin an antiplatelet?
Aspirin reduces the risk of serious vascular events in patients at high risk of such an event by about a quarter and is recommended as the first-line antiplatelet drug. Clopidogrel reduces the risk of serious vascular events among high-risk patients by about 10% compared with aspirin.
What functional groups Does aspirin have?There are three different functional groups in aspirin, which contribute to its properties, a weak acid soluble in basic solutions. These functional groups include carboxylic acid, ester, and an aromatic group.
Article first time published onIs aspirin a methyl salicylate?
19.2 Methyl Salicylate Poisoning Although aspirin is acetyl salicylate that is structurally close to methyl salicylate, after ingestion it is rapidly broken down into salicylic acid by blood and liver esterase; very little methyl salicylate is broken down to salicylic acid by the esterase in blood.
What ester is found in aspirin?
Aspirin is a trade name for acetylsalicylic acid, a common analgesic. Acetylsalicylic acid is an acetic acid ester derivative of salicylic acid.
Which functional group is found in an ester?
ester: A compound most often formed by the condensation of an alcohol and an acid, with elimination of water. It contains the functional group C=O. joined via carbon to another oxygen atom. ether: Compound containing an oxygen atom bonded to two hydrocarbon groups.
Is aspirin soluble in hexane?
Acetylsalicylic acid is fairly polar and not soluble in pure hexane. Ethyl acetate (CH3CO2CH2CH3) is much more polar and your compound is very soluble in pure ethyl acetate.
Is aspirin soluble in ethanol?
Aspirin is soluble in organic solvents such as ethanol, DMSO, and dimethyl formamide, which should be purged with an inert gas. … The solubility of aspirin in PBS, pH 7.2, is approximately 2.7 mg/ml.
Why is aspirin insoluble in water?
Aspirin, or acetylsalicylic acid, or 2-hydroxybenzoic acid 2 carboxyphenyl ester, has a benzene ring, (C6H6) which is hydrophobic (water-hating), and this portion of the molecule does not interact with water.
Is aspirin a polar?
Aspirin is a polar molecule with dipole-dipole attraction bonds and an -OH (hydroxyl) segment as part of a carboxylic acid group.
What is aspirin chemistry?
One of the best known aromatic acetates is acetylsalicylic acid, or aspirin, which is prepared by the esterification of the phenolic hydroxyl group of salicylic acid. Aspirin possesses a number of properties that make it the most often recommended drug. It is an analgesic, effective in pain relief.
Does aspirin have a ketone group?
The male sex hormone testosterone contains ketone, alkene, and secondary alcohol groups, while acetylsalicylic acid (aspirin) contains aromatic, carboxylic acid, and ester groups.
Is aspirin a carboxylic acid?
Aspirin is both an aromatic carboxylic acid (red oval) and a phenyl ester of acetic acid (blue oval).
How aspirin is metabolized?
Therefore, aspirin has a very short half-life. Salicylate, in turn, is mainly metabolized by the liver. This metabolism occurs primarily by hepatic conjugation with glycin or glucuronic acid, each involving different metabolic pathways. The predominant pathway is the conjugation with glycin, which is saturable.
How do you make aspirin a level chemistry?
- Place 2.0 g (0.015 mole) of salicylic acid in a 125-mL Erlenmeyer flask.
- Add 5 mL (0.05 mole) of acetic anhydride, followed by 5 drops of conc. …
- Heat the flask gently on the steam bath for at least 10 minutes.
- Allow the flask to cool to room temperature.
Is aspirin selective or nonselective?
COX inhibitors divide into non-selective nonsteroidal anti-inflammatory drugs (NSAIDs), COX-2 selective nonsteroidal anti-inflammatory drugs (c2s NSAIDs), and aspirin. NSAIDs include ibuprofen, naproxen, ketorolac, and indomethacin.
Is aspirin an antagonist or agonist?
Aspirin and P2Y12 antagonists are commonly used anti-platelet agents. Aspirin produces its effects through inhibition of thromboxane A2 (TXA2) production, while P2Y12 antagonists attenuate the secondary responses to ADP released by activated platelets.
Is aspirin a anticoagulant or antiplatelet?
There are two main types of blood thinners. Anticoagulants such as heparin or warfarin (also called Coumadin) slow down your body’s process of making clots. Antiplatelet drugs, such as aspirin, prevent blood cells called platelets from clumping together to form a clot.
Is aspirin a benzene?
Benzene is one of the most widely used chemicals in the US today. It is used predominately as a starting material in making other chemicals–including aspirin and other drugs.
Is there benzene in aspirin?
In fact, the starting material for the chemical synthesis of aspirin is benzene, derived from petroleum. This is then converted to phenol which in turn is converted to salicylic acid which is then converted to acetylsalicylic acid or ASA, which we know as aspirin.
What Tree Is aspirin from?
The story of the discovery of aspirin stretches back more than 3500 years to when bark from the willow tree was used as a pain reliever and antipyretic. It involves an Oxfordshire clergyman, scientists at a German dye manufacturer, a Nobel Prize-winning discovery and a series of pivotal clinical trials.
Which functional group is found in an ester quizlet?
esters are chemical compounds derived from an acid (organic or inorganic) in which at least one -OH (hydroxyl) group is replaced by an -O-alkyl (alkoxy) group.
What is the primary function of aspirin?
Aspirin is a common drug for relieving minor aches, pains, and fevers. People also use it as an anti-inflammatory or a blood thinner. People can buy aspirin over the counter without a prescription. Everyday uses include relieving headache, reducing swelling, and reducing a fever.
What functional group in aspirin irritates the stomach?
It is the blocking of COX-1 that is responsible for the increased risk of stomach irritation when aspirin is taken, as it stops the formation of a protective prostaglandin (prostacyclin), in cells in the stomach lining, which reduces gastric acid production.