What is base excess in arterial blood gases
It is defined as the amount of acid required to restore a litre of blood to its normal pH at a PaCO2 of 40 mmHg. The base excess increases in metabolic alkalosis and decreases (or becomes more negative) in metabolic acidosis, but its utility in interpreting blood gas results is controversial.
What does base excess measure?
The base excess (BE) is a way to quantify the presence of strong acid (metabolic acidosis) or strong base (metabolic alkalosis), either (1) in the whole blood, called blood base excess, BE(B), or actual base excess, ABE, or (2) in the extracellular fluid, called extracellular BE (BEecf) or standard BE (SBE).
Is base deficit the same as base excess?
A positive number is called a base excess and indicates a metabolic alkalosis. A negative number is called a base deficit and indicates a metabolic acidosis.
What is a normal base excess?
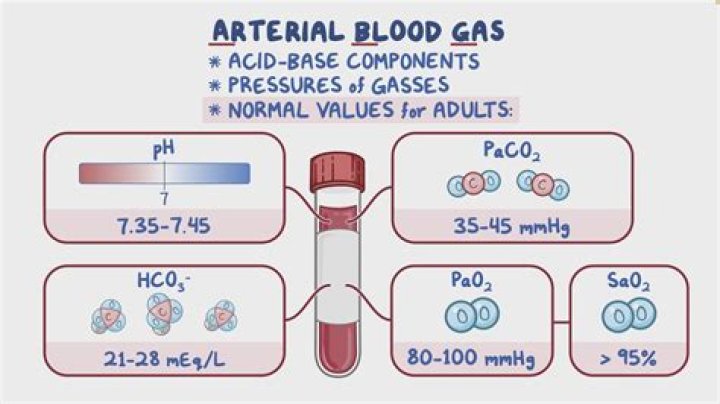
base excess is dose of acid or alkali to return in vitro blood to normal pH (7.40) under standard conditions ( at 37C at a PCO2 of 40 mm Hg) Normal Base excess is between -3 and +3mEq/L.How is base excess calculated?
Most equations used for calculation of the base excess (BE, mmol/l) in human blood are based on the fundamental equation derived by Siggaard-Andersen and called the Van Slyke equation: BE = Z x [[cHCO3-(P) – C7. 4 HCO3-(P)] + beta x (pH -7.4)].
What affects base excess?
Base excessLOINC11555-0
What causes high base excess?
A high base excess (> +2mmol/L) indicates that there is a higher than normal amount of HCO3– in the blood, which may be due to a primary metabolic alkalosis or a compensated respiratory acidosis.
What causes respiratory acidosis?
Respiratory acidosis typically occurs due to failure of ventilation and accumulation of carbon dioxide. The primary disturbance is an elevated arterial partial pressure of carbon dioxide (pCO2) and a decreased ratio of arterial bicarbonate to arterial pCO2, which results in a decrease in the pH of the blood.Is base deficit higher when pH is high?
BG ParameterUmbilical ArteryUmbilical VeinBase Deficit+9.3 to -1.5+8.3 to -2.6
Which conditions can cause metabolic acidosis?- Cancer.
- Carbon monoxide poisoning.
- Drinking too much alcohol.
- Exercising vigorously for a very long time.
- Liver failure.
- Low blood sugar (hypoglycemia)
- Medicines, such as salicylates, metformin, anti-retrovirals.
- MELAS (a very rare genetic mitochondrial disorder that affects energy production)
How do you determine base deficit?
The arterial base deficit (BD) is directly calculated from the blood gas analyzer from the PCO2, pH, and serum bicarbonate (HCO3) values as applied to a standard nomogram and represents the number of milliequivalents of additional base that must be added to a liter of blood to normalize the pH.
How do you interpret an ABG?
If the ABG results reveal pH numbers are not within the normal range, the patient’s pH level is either acidotic or alkalotic. The lower the number, the more acidotic the patient is. For instance, a pH of 3 is severely acidotic and requires emergency intervention. Alkalosis is the opposite.
How do you correct metabolic acidosis?
- insulin.
- diabetes medications.
- fluids.
- electrolytes (sodium, chloride, potassium)
What is normal bicarb?
Normal bicarbonate levels are: 23 to 30 mEq/L in adults.
How do you fix respiratory alkalosis?
- Breathe into a paper bag. Fill the paper bag with carbon dioxide by exhaling into it. …
- Get reassurance. The symptoms of respiratory alkalosis can be frightening. …
- Restrict oxygen intake into the lungs. To do this, try breathing while pursing the lips or breathing through one nostril.
How do you fix metabolic alkalosis?
- Saline infusion.
- Potassium replacement.
- Magnesium replacement.
- Chloride infusion.
- Hydrochloric acid infusion.
- Stopping the medications that caused the condition, for example high doses of diuretics.
What is acid base disorders?
Acid-base disorders are pathologic changes in carbon dioxide partial pressure (Pco 2) or serum bicarbonate (HCO 3 −) that typically produce abnormal arterial pH values. Acidemia is serum pH < 7.35. Alkalemia is serum pH > 7.45. Acidosis refers to physiologic processes that cause acid accumulation or alkali loss.
Which is the most predominant buffer system in the body?
Because of its high concentration in blood, the bicarbonate/carbonic acid pair is the most important buffer system in the blood. This buffer system is also effective in the lungs and in the kidneys in helping to regulate body ph.
What is base deficit in trauma?
In trauma patients, arterial base deficit shows tissue injury severity, because it is correlated with arterial lactate concentration. Elevated base deficit and lactate concentrations after shock are related to oxygen transport imbalance at the cellular level.
What is a normal cord pH?
The normal (reference) range for arterial cord blood pH is 7.12-7.35, and for arterial cord BD it is +9.3 to –1.5 mmol/L. In obstetrics, metabolic acidosis is most widely defined as cord arterial blood pH <7.0 and BD >12.0 mmol/L, although some suggest the threshold pH should be higher (<7.1).
What happens in the body during respiratory acidosis?
Respiratory acidosis is a condition that occurs when the lungs can’t remove enough of the carbon dioxide (CO2) produced by the body. Excess CO2 causes the pH of blood and other bodily fluids to decrease, making them too acidic. Normally, the body is able to balance the ions that control acidity.
What does too much CO2 in the blood cause?
Having too much carbon dioxide in the body can cause symptoms like headache, fatigue, and muscle twitches—such common ailments that you can see why many people dismiss the symptoms as simply pesky. Most of the time, they are.
What lab values indicate respiratory acidosis?
Respiratory Acidosis pH<7.35. HCO3- > 28 mEq/L (if compensating) PaCO2 > 45 mm Hg.
Is CO2 high or low in metabolic acidosis?
For a respiratory acidosis, the pCO2 is greater than 40 to 45 due to decreased ventilation. Metabolic acidosis is due to alterations in bicarbonate, so the pCO2 is less than 40 since it is not the cause of the primary acid-base disturbance.
What are signs of acidosis?
- rapid and shallow breathing.
- confusion.
- fatigue.
- headache.
- sleepiness.
- lack of appetite.
- jaundice.
- increased heart rate.
What is the difference between metabolic acidosis and respiratory acidosis?
Acidosis that occurs when the lungs fail to remove excess carbon dioxide from our bloodstream during the process of respiration is respiratory acidosis. Acidosis that occurs when the digestive and urinary systems fail to breakdown and maintain the proper level of acids in the blood is known as metabolic acidosis.
What is base deficit in metabolic acidosis?
Base deficit is the amount of base (in mmol) required to titrate a liter of whole arterial blood to a pH of 7.40. Base deficit as a measure of metabolic acidosis was first proposed by Anderson and Engel in 1960[1].
What pH value of an arterial blood gas sample indicates respiratory failure?
PaCO2 > 40 with a pH < 7.4 indicates a respiratory acidosis, while PaCO2 < 40 and pH > 7.4 indicates a respiratory alkalosis (but is often from hyperventilation from anxiety or compensation for a metabolic acidosis).
How do you know if a mixture is acid base?
To identify mixed acid-base disorders, blood gas analysis is used to identify primary acid-base disturbance and determine if an appropriate compensatory response has developed. Inappropriate compensatory responses (inadequate or excessive) are evidence of a mixed respiratory and metabolic disorder.
How do you know if ABG is metabolic or respiratory?
- Use pH to determine Acidosis or Alkalosis. ph. < 7.35. 7.35-7.45. …
- Use PaCO2 to determine respiratory effect. PaCO2. < 35. …
- Assume metabolic cause when respiratory is ruled out. You’ll be right most of the time if you remember this simple table: High pH. …
- Use HC03 to verify metabolic effect. Normal HCO3- is 22-26. Please note:
What are the 6 steps to ABG analysis?
- Analyze the pH. …
- Analyze the PaCO2. …
- Analyze the HCO3. …
- Match either the PaCO2 or the HCO3 with the pH. …
- Determine whether the PaCO2 or the HCO3 go in the opposite direction of the pH. …
- Analyze the PaO2 and SaO2 for hypoxemia.



