What is the electron pair geometry of the Se atom in SeF4
As per the VSEPR chart, if a molecule central atom is attached with four bonded atoms and has one lone pair then the molecular geometry of that molecule is see-saw in nature, and electron geometry is trigonal bipyramidal. Hence, the molecular shape for SeF4 is see-saw and its electron geometry is trigonal bipyramidal.
What is the molecular geometry of SE?
The electron geometry around Se is trigonal planar.
What is the electron pair of geometry?
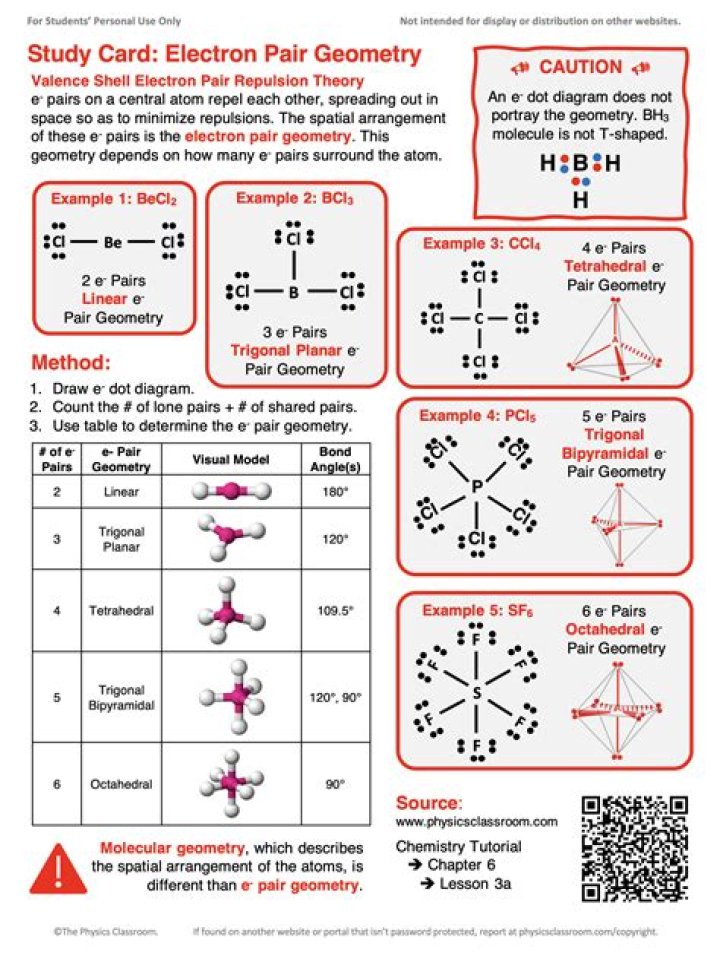
electron-pair geometry: arrangement around a central atom of all regions of electron density (bonds, lone pairs, or unpaired electrons) linear: shape in which two outside groups are placed on opposite sides of a central atom. molecular structure: structure that includes only the placement of the atoms in the molecule.
Is SeF4 tetrahedral?
This means that SeF4 has Trigonal Bipyramidal structure with 4 bond pairs and 1 lone pair.How many lone pairs are on se?
Based on the structure, there are two lone pairs in selenium. Therefore, the answer is B.
Is SeF4 a geometry?
SeF4 is a polar molecule because of asymmetrical geometry that causes the non-uniform distribution of charge in the molecule. In the SeF4 lewis structure, a total of 13 lone pairs and 4 bond pairs are present. The molecular geometry of SeF4 is see-saw and electron geometry is trigonal bipyramidal.
Is n20 linear?
Nitrous oxide is linear.
Is SeF4 polar or non polar?
Yes,selenium tetrafluoride that means SeF4 is a polar molecule because it is of AB4E type molecule. Hence it’s geometry is trigonal bipyramidal and shape is see saw. here net dipole moment does not equal to zero. so that given molecule is polar.What was the molecular geometry for scl6?
The electron pair geometry and molecular geometry are both octahedral. All bond angles are 90⁰. An example is sulfur hexachloride (SCl6).
How do you find electron geometry?- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. …
- Name the electron-group geometry. …
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.
Is electron geometry and molecular geometry the same?
The molecular geometry definition in chemistry is the arrangement of atoms in relation to a central atom in three-dimensional space. Electron geometry is the arrangement of electron groups. … If all the electron groups are bonded, with no lone pairs, then the electron geometry and molecular geometry are the same.
How many lone pairs of electrons are on the SE atom SeCl2?
Question: In the hypothetical molecule SeCl2, the central atom Se has two lone pairs of electrons in addition to the two bond pairs in the Se-cl bonds.
How many lone pairs are in CL?
Each Cl atom is surrounded by three (3) lone pairs and shares one lone pair. This makes a total of eight (8) valence electrons surrounding each Cl atom.
How many lone pairs of electrons are on the SeCl2?
due to more repulsions between lone pair-lone pair, all 3 lone pairs are located in equitorial position and axial line is located with 2 bond pairs of chlorine.
Is N2O a geometry?
N2O molecular geometry is a linear shape with a bond angle of 180 degrees. Nitrogen(N) and Oxygen(O) atoms are covalently linked in the N2O molecular geometry. N and O have five and six valence electrons, respectively. N2O contains a total of 16 valence electrons.
Is n20 trigonal planar?
Nitrogen Dioxide: The nitrogen and and one oxygen are bonded through a double bond which counts as “one electron pair”. Hence the molecule has three electron pairs and is trigonal planar for electron pair geometry.
What is the geometry of N2O 5?
NamesCrystal structurehexagonalMolecular shapeplanar, C2v (approx. D2h) N–O–N ≈ 180°ThermochemistryStd molar entropy (So298)178.2 J K−1 mol−1 (s) 355.6 J K−1 mol−1 (g)
What is the electron domain geometry of sebr4?
tetrahedral see-saw square planar square pvramidal trigonal pyramidal.
How many electron domains does SeF4?
234 and five. So we have a total of five domains.
Does scl6 have polar molecules?
Step 1: Sulfur (EN = 2.5) is less electronegative than chlorine (EN = 3) so sulfur is the central atom. … Step 5: We can see that the dipole arrows from S–Cl bond points in the opposite direction so they cancel out. This means there is a no net dipole in the molecule, making it non-polar.
Is SE4 polar?
The SE Electronics SE4 is a cardioid “pencil” style microphone designed to capture instruments and vocals in sound reinforcement and recording applications. … The microphone’s cardioid polar pattern is effective in minimizing noise and unwanted audio signal at the off-axis sections of the microphone.
Does SeF4 violate the octet rule?
From the structure “Se” have four bonds. And central atom “ Se” surrounded by eight bonding electrons and one pair of nonbonding electrons . Hence ,SeF4 does not obey the octet rule.
What is the electron pair geometry for O in h2o?
VSEPR calculation for water, OH. Water has four electron pairs and the coordination geometry of oxygen is based upon a tetrahedral arrangement of electron pairs.
What is the electron geometry if you have 2 electron groups around the center atom?
30) It is important to identify lone pairs around a central atom because they do not repel other electron groups. 31) The electron geometry of a molecule is never the same as its molecular geometry. 32) When you have 2 electron groups and none are lone pairs, the molecular geometry is linear.
What shape is a tetrahedral?
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners.
What is the electron pair geometry for a molecule with three electron pairs around the central atom?
# of bonding groups/domains on ‘central’ atom# of lone pair electrons on ‘central’ atomMolecular Geometry30trigonal planar21bent40tetrahedral31trigonal pyramidal
What is the bond angle for SeCl2?
The bond distance in SeCl2 is ra(Se-Cl) = 2.157(3) Ä, the valence angle <ClSeCl = 99.6(5)°.
How many lone pairs are in ph3?
Phosphorus forms three bond pairs and one lone pair.
How many pairs of electrons are in Chloride?
Explanation: An isolated chlorine ATOM has 7 valence electrons. And thus the parent dichlorine molecule tends to be an OXIDANT, viz…… And because the resultant chloride anion has FOUR SUCH LONE PAIRS, instead of the SEVEN electrons associated with the NEUTRAL atom, chloride has a FORMAL NEGATIVE CHARGE.



