What is the valence shell of oxygen
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. We can write the configuration of oxygen’s valence electrons as 2s²2p⁴.
What is the shell of oxygen?
ZElementNo. of electrons/shell5Boron2, 36Carbon2, 47Nitrogen2, 58Oxygen2, 6
What is the valence shell number?
Valence shell is the outermost shell of every element. Atom of every element have different electronic configurations based on the atomic number of each element. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. For example, Sodium.
What is a valence shell?
Definition of valence shell : the outermost shell of an atom containing the valence electrons.How many shells does oxygen have?
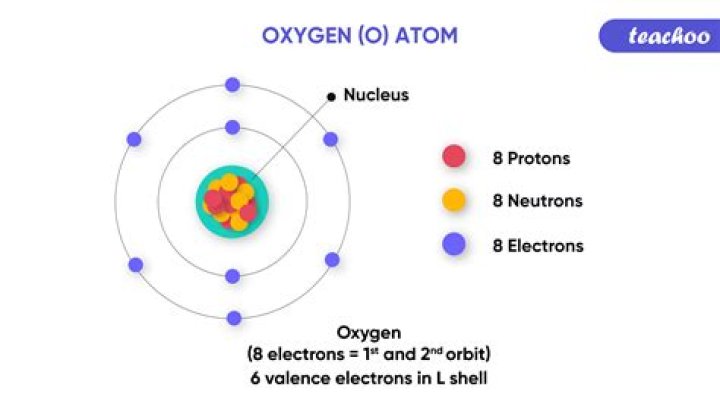
So… for the element of OXYGEN, you already know that the atomic number tells you the number of electrons. That means there are 8 electrons in an oxygen atom. Looking at the picture, you can see there are two electrons in shell one and six in shell two.
What do you mean by L-shell?
Definition of L-shell : the second innermost shell of electrons surrounding an atomic nucleus — compare k-shell , m-shell.
What is another way an oxygen to fill its valence shell?
Oxygen, which has six valence electrons, must participate in two covalent bonds to complete its octet. This means that oxygen retains two pairs of non-bonded electrons that are not shared with any other atom.
What is valence shell Class 10?
What are Valence Electrons? Valence is the number of electrons an atom must lose or gain to attain the nearest noble gas or inert gas electronic configuration. “Electrons in the outer shells that are not filled are called valence electrons”.What is valence shell and valence electron?
Valence shell: The outermost orbital shell of an atom is called its valence shell. These electrons take part in bonding with other atoms. Valence electrons: Electrons in the outer shells that are not filled are called valence electrons.
How many valence E does CL have?A: An atom of a group 17 element such as chlorine has seven valence electrons. It is “eager” to gain an extra electron to fill its outer energy level and gain stability.
Article first time published onWhy does oxygen have 2 valence electrons?
Oxygen needs 2 more electrons to fill its outermost shell. … In order to complete its octet, it needs 8 electrons in its outermost shell. Since it already has 6, it would only require 2 more electrons to complete its octet.
What is the outermost shell of oxygen?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. We can write the configuration of oxygen’s valence electrons as 2s²2p⁴.
Why are full valence shells stable?
A complete octet is very stable because all orbitals will be full. Atoms with greater stability have less energy, so a reaction that increases the stability of the atoms will release energy in the form of heat or light. A stable arrangement is attended when the atom is surrounded by eight electrons.
How many valence does nitrogen have?
Nitrogen is found to have either 3 or 5 valence electrons and lies at the top of Group 15 on the periodic table. It can have either 3 or 5 valence electrons because it can bond in the outer 2p and 2s orbitals.
How many electrons N shell contains?
Answer: N shell can have a maximum of 32 electrons.
How many orbitals are in the L shell?
Answer: There is one orbital in an s subshell (l = 0), three orbitals in a p subshell (l = 1), and five orbitals in a d subshell (l = 2).
What is the valence shell of magnesium?
Magnesium has atomic number 12. Its electronic configuration is 2,8,2 . Thus, it has 2 valence electrons.
What is the valence shell last shell of the elements in Group IIA?
The columns indicate, for the neutral form of the element, how many valance electrons are in the valance shell: the first column (Group IA) contains 1 valence electron, the second column (Group IIA) contains 2 valence electrons, etc., and the last column (Group VIIIA) contains 8 valence electrons.
Why the outermost shell is called valence shell?
The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons.
Is valence shell and outermost shell same?
The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons.
What is valence shell Class 11?
Valence shell is the outermost shell of an atom that contains electrons. … The electrons in these valence shells are called valence electrons. Example :- Sodium metal has atomic number 11.
What is Prepenultimate shell?
The electrons are arranged in an atom in the various shells around the nucleus. The last shell or the outer most shell from the nucleus is called the valence shell. The shell inner to this is called the penultimate shell. and the one inner to penultimate shell is called the prepenultimate or anti-penultimate shell.
What is valence electron Class 9?
The outermost electron shell of an atom is called valence shell. The electrons present in outermost shell of an atom are called as valence electrons. The valence electron of an atom take part in a chemical reaction because they have more energy than all the inner electrons.
What is the valence of chlorine?
CompoundFormulaValenceChlorineCl2Cl = 1Hydrogen peroxideH2O2H = 1 O = 2AcetyleneC2H2C = 4 H = 1Mercury(I) chlorideHg2Cl2Hg = 2 Cl = 1
How do you find the valence of an element?
For neutral atoms, the number of valence electrons is equal to the atom’s main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons. Oxygen is in group 6 and has 6 valence electrons.
Is Cl A metal?
Chlorine is a non-metal. … The element chlorine is a yellowish-green gas which is extremely reactive and react with various metals.
What group is chlorine in?
Chlorine is in group 17 of periodic table, also called the halogens, and is not found as the element in nature – only as a compound. The most common of these being salt, or sodium chloride, and the potassium compounds sylvite (or potassium chloride) and carnallite (potassium magnesium chloride hexahydrate).
How many valence shell electrons does boron have?
The boron atom has only six electrons in its outer shell, leading to an electron deficiency. This molecule has 12 valence shell electrons; 3 each from the B atoms, and 1 each from the six H atoms.



