Why is fluorine a bad leaving group
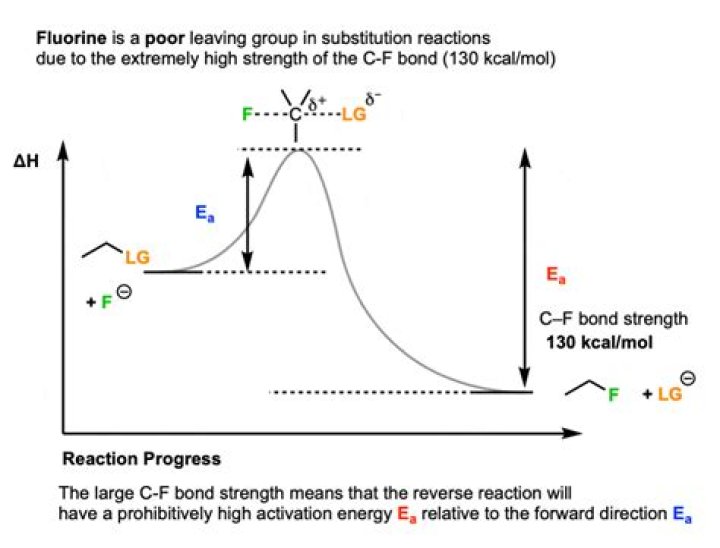
Exception: Fluorine is a poor leaving group. F⁻ is a small ion. Its high charge density makes it relatively unpolarizable. The leaving group needs to be polarizable to lower the energy of the transition state.
Is Fluorine is a good leaving group?
Fluorine tends to be a very poor leaving group for SN1/SN2/E1/E2 reactions. In Org 2, you may see some examples where F can act as a leaving group when it is attached to a carbonyl carbon or an aromatic ring.
Is fluorine or chlorine a better leaving group?
Fluorine is not a good leaving group, but that doesn’t matter as I said before. It is not the leaving group ability of F− which makes the reaction go faster than with, say, bromine or chlorine, but its very high negative inductive effect (due to its large electronegativity).
What makes a bad leaving group?
Some examples of weak bases: halide ions (I-, Br-, Cl-) water (OH2), and sulfonates such as p-toluenesulfonate (OTs) and methanesulfonate (OMs). The weaker the base, the better the leaving group. On the other hand, strong bases are bad leaving groups.Why is bromine better leaving group than fluorine?
The large size of the bromine is able to dilute the negative charge, which has a stabilizing effect, while the smaller fluorine atom’s negative charge will be more concentrated and therefore less stable.
Why is hydroxyl a bad leaving group?
Alcohols have hydroxyl groups (OH) which are not good leaving groups. Why not? Because good leaving groups are weak bases, and the hydroxide ion (HO–) is a strong base. … This will convert the alcohol into an alkyl bromide or alkyl chloride, respectively, and halides (being weak bases) are great leaving groups.
Why is Alkoxide a poor leaving group?
Let us examine the basic, SN2 case first. The leaving group is an alkoxide anion, because there is no acid available to protonate the oxygen prior to ring opening. An alkoxide is a poor leaving group, and thus the ring is unlikely to open without a ‘push’ from the nucleophile.
Which is better leaving group?
Typically the leaving group is an anion (e.g. Cl-) or a neutral molecule (e.g. H2O). The better the leaving group, the more likely it is to depart. A “good” leaving group can be recognised as being the conjugate base of a strong acid.Is h2so4 a good leaving group?
As an anion, its stability and appearance are very similar to the bisulfate conjugate base of strongly acidic sulfuric acid (see below), so it is not surprising that it is such a good leaving group and stable conjugate base.
Is ts a good leaving group?The tosylate group makes for an excellent leaving group and is commonly used in organic chemistry in a multitude of reactions. … This is a most stabilizing effect and this is what makes the tosylate group such a good leaving group.
Article first time published onWhich Halogen is the best leaving group?
Iodide, which is the least basic of the four common halides (F, Cl, Br, and I), is the best leaving group among them. Fluoride is the least effective leaving group among the halides, because fluoride anion is the most basic.
Why is fluorine a bad Nucleophile?
As fluoride ion has the greatest number of lone pairs (= 4), it should suffer from highest number of lone pair-lone pair repulsions. In order to reduce its lone pair-lone pair repulsions and thus its unstability, shouldn’t it donate its lone pair more readily than other anions?
Is NH2 a good leaving group?
Water is neutral whereas ammonia is a weak base (even less acidic). Stronger conjugate acids have weaker conjugate base and vice versa . Therefore, NH2- is a stronger base. OH- is better leaving grp because NH2- is stronger base.
Why is iodide a good leaving group?
The I- ion is a great leaving group due to its large atomic radius, meaning that the x-I bond (where x is an atom bonded to the iodine in a specific compound) is weaker (has a lower bond enthalpy, as it is longer and there are less electrostatic forces of attraction between the two positive nuclei and the shared Pair …
What is leaving group ability?
The physical manifestation of leaving group ability is the rate at which a reaction takes place. Good leaving groups give fast reactions. By transition state theory, this implies that reactions involving good leaving groups have low activation barriers leading to relatively stable transition states.
Is OET a good leaving group?
3 Answers. The alkoxide and the hydroxides aren’t good leaving groups. Consider an alcohol, the OH group never leaves on its own.
Is ammonia a good leaving group?
The difference between the eliminations of alcohols and amines in acidic solution is the poorer leaving group ability of ammonia than that of water (remember, ammonia is a stronger base; therefore a poorer leaving group.)
Which is a better leaving group tosylate or mesylate?
, mesylate and tosylate are better leaving groups than alcohols are. … Usually, the best leaving groups are very weak bases. Methanesulfonic acid and p-toluenesulfonic acid are extremely strong acids, Their conjugate bases are extremely weak, so the conjugate bases are excellent leaving groups.
What does h2so4 do to an alkene?
Alkenes react with concentrated sulfuric acid in the cold to produce alkyl hydrogensulphates. For example, ethene reacts to give ethyl hydrogensulphate. The structure of the product molecule is sometimes written as CH3CH2HSO4, b ut the version in the equation is better because it shows how all the atoms are linked up.
What does h2so4 do as a reagent?
Because sulfuric acid is also a strong oxidizing agent, it oxidizes some of the alcohol to carbon dioxide and is simultaneously reduced itself to sulfur dioxide. Both of these gases must be removed from the alkene. Sulfuric acid also reacts with the alcohol to produce a mass of carbon.
What happens when an alcohol is heated with h2so4?
When ethanol is heated with concentrated sulphuric acid at 170oC, it gets dehydrated to form ethene (unsaturated hydrocarbon). This reaction is termed as dehydration reaction as it involves the removal of water molecules from the alcohol (ethanol).
Why do leaving groups leave?
In order for a leaving group to leave, it must be able to accept electrons. A strong bases wants to donate electrons; therefore, the leaving group must be a weak base.
How does the leaving group affect SN1 reactions?
An SN1 reaction speeds up with a good leaving group. This is because the leaving group is involved in the rate-determining step. A good leaving group wants to leave so it breaks the C-Leaving Group bond faster. … This is because the better leaving group leaves faster and thus the reaction can proceed faster.
What is leaving group in organic chemistry?
Leaving group (LG; nucleofuge): An atom or group of atoms which breaks away from the rest of the molecule, taking with it the electron pair which used to be the bond between the leaving group and the rest of the molecule. … Bromide ion is the leaving group in this SN2 reaction.
Is tosylate a better leaving group than bromide?
Tosylate is a better leaving group than bromide. Preparation of tosylate does not affect any of the bonds to the stereogenic center, so configuration and optical purity of tosylate is the same as the alcohol from which it was formed.
What does TsCl do to an alcohol?
We use “mesyl chloride” (MsCl) or “tosyl chloride” (TsCl), and the neutral alcohol performs a substitution reaction on sulfur, leading to formation of O-S and breakage of S-Cl. Then, deprotonation of the charged alcohol leads to the neutral mesylate or tosylate.
Is Meoh a good leaving group?
Good for Sn1, not the best for Sn2. 4) Leaving group: Br –, a very good leaving group. Decision: … Methanol is a polar protic solvent, which is good for a Sn1 reaction.
Is iodine or fluorine a better leaving group?
Weaker bases are better leaving groups Iodide, which is the least basic of the four common halides (F, Cl, Br, and I), is the best leaving group among them. Fluoride is the least effective leaving group among the halides, because fluoride anion is the most basic.
Why is chloride a good leaving group?
Chlorides, bromides, and tosylate / mesylate groups are excellent leaving groups in nucleophilic substitution reactions, due to resonance delocalization of the developing negative charge on the leaving oxygen.
Is fluorine nucleophilic?
As can be seen, the fluoride ion is one of a number of highly basic nucleophiles whose basicity sharply decreases in aprotic and especially protic media.
Why is H2S a better nucleophile than H2O?
According to my notes, nucleophilicity order is directly proportional to basicity order in an aprotic medium, and is inversely proportional to basicity order in protic medium. And H2O is a stronger base than H2S, so it should be the strong nucleophile, but the answer is given as H2S is the stronger nucleophile.



